Understanding Charles’ Law and Gas Volume
Discover Jacques Charles' findings on the relationship between volume and temperature in gases through his experiments with hot air balloons. Learn how the volume of a gas changes with temperature variations at constant pressure, in-line with Charles’ Law. Explore the direct relationship between temperature and volume, explained by kinetic molecular theory equations.

Understanding Charles’ Law and Gas Volume
E N D
Presentation Transcript
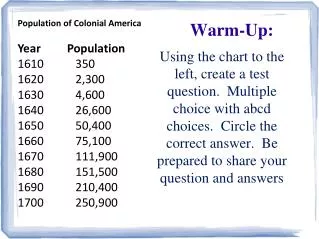
Warm-Up A gas is pumped into a container with a volume of 2.8L at 5.63atm. If the pressure is increased to 25.3atm, what will the new volume be?
Jacques Charles • 1746-1823 • French chemist • Discovered the relationship between volume and temperature while experimenting with hot air balloons.
Charles’ Law • At constant pressure, temperature and volume have a direct relationship. • As one goes up the other goes up, too. • Can be explained through kinetic molecular theory. • Increasing temperature increases average kinetic energy. This causes the molecules to move faster and collide the container with more force. To counter this force, the volume will increase.
V1 = V2T1 T2 • V1= initial volume • T1= initial temperature* • V2= final volume • T2= final temperature* *temperature must be in Kelvin scale! K= °C+273
A gas has a volume of 2.5 L at 35.°C. What would the new volume be if the temperature was raised to 100.°C? V1 = V2T1 T2 2.5 = V2308 373 932.5 = 308V2 308 3.0L= V2
A sample of neon gas occupies a volume of 752 mL at 25°C. What volume will the gas occupy at 50°C if the pressure remains constant? V1 = V2T1 T2 752 = V2298 323 242896 = 298V2 298 298 815mL= V2
A gas at 65°C occupies 4.22 L. At what temperature will the volume be 3.87 L, assuming the same pressure? V1 = V2T1 T2 4.22 = 3.87 338 T2 1308.06 = 4.22T2 4.22 4.22 310K= T2