Chemical Reactions Balancing and Types Guide
Learn how to balance chemical reactions and identify different types including combinations, decompositions, single and double replacements, and combustions with examples. Explore rules for predicting products and complete equations effortlessly.

Chemical Reactions Balancing and Types Guide
E N D
Presentation Transcript
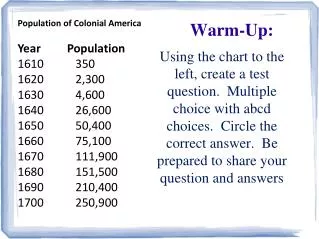
Warm Up Balance the following reactions: • __NaCl + __BeF2 --> __NaF + __BeCl2 2. __FeCl3 + __Be3(PO4)2 --> __BeCl2 + __FePO4
Warm Up Balance the following reactions: • _2_NaCl + __BeF2 --> _2_NaF + __BeCl2 2. 2_FeCl3 + _Be3(PO4)2 --> _3BeCl2 + _2FePO4
Combination Reactions A + B AB Examples: S + O2 SO2 CaO + H2O → Ca(OH)2
Decomposition Reactions AB A + B Examples 2H2O(I) → 2H2 + O2 2Cl2O5 (g) → 2Cl2 (g) + 5O2 (g)
Single Replacement Reaction A + BC → AC + B Example: Cu + 2AgNO3 → 2Ag + Cu(NO3)2 Br2 + 2KI → 2KBr + I2
Double Replacement Reactions AX + BY → BX + AY Example: HCl (aq) + NaOH (aq) → NaCl (aq) + H2O (l)
Combustion Reaction • An element or compound reacts with an oxidizing element, such as oxygen. • Probable products are CO2 and H2O Examples: CH4 + 2 O2 → CO2 + 2 H2O + energy 2 H2 + O2 → 2 H2O(g) + heat
Rules for Predicting Products • R+ S RS • RS R + S • T + RS TS + R • R+S- + T+U- R+U- + T+S- • CxHy + (x+y/4) O2 x CO2 + (y/2) H2O
Reminder Make sure that you look at what might be formed and consider its state when predicting products • Metals will form solids on their own and be ions in combination with nonmetals Mg compared to MgCl2 • Don’t forget that some non-metals will be anions in combination with metals, but covalent diatomic gasses when they are on their “own” Cl2 compared to HCl
Complete the following equations: Decomposition • NCl3 Single Replacement • Al + CuSO4 • Mg + HCl Double Replacement • Ba(NO3)2 + Na2SO4 *** Hint Nitrogen and Chlorine are diatomic gasses on their own ** *** Hint: Look at the ions you are working with!!!***
Complete the following equations: Decomposition • NCl3 N2 + Cl2 Single Replacement • Al + CuSO4 • Mg + HCl Double Replacement • Ba(NO3)2 + Na2SO4
Complete the following equations: Decomposition • NCl3 N2 + Cl2 Single Replacement • Al + CuSO4 Al2(SO4)3 + Cu • Mg + HCl Double Replacement • Ba(NO3)2 + Na2SO4
Complete the following equations: Decomposition • NCl3 N2 + Cl2 Single Replacement • Al + CuSO4 Al2(SO4)3 + Cu • Mg + HCl H2 + MgCl2 Double Replacement • Ba(NO3)2 + Na2SO4
Complete the following equations: Decomposition • NCl3 N2 + Cl2 Single Replacement • Al + CuSO4 Al2(SO4)3 + Cu • Mg + HCl H2 + MgCl2 Double Replacement • Ba(NO3)2 + Na2SO4 BaSO4 + NaNO3
The End Start Working on HW if time permits