Warm-up
130 likes | 780 Views
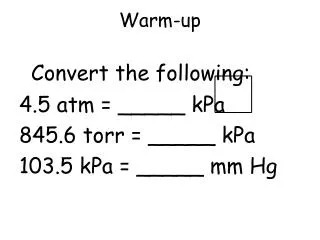
Warm-up. Convert the following: 4.5 atm = _____ kPa 845.6 torr = _____ kPa 103.5 kPa = _____ mm Hg. Boyle’s Law: P and V. as one increases , the other decreases inversely proportional P 1 = P 2 V 2 V 1. Example: Boyle’s Law.

Warm-up
E N D
Presentation Transcript
Warm-up Convert the following: 4.5 atm = _____ kPa 845.6 torr = _____ kPa 103.5 kPa = _____ mm Hg
Boyle’s Law: P and V • as one increases, • the other decreases • inversely proportional P1 = P2 V2 V1
Example: Boyle’s Law Consider a 1.53-L sample of gaseous SO2 at a pressure of 5.6 x 103 Pa. If the pressure is changed to 1.5 x 104 Pa at constant temperature, what will be the new volume of the gas?
Charles’ Law: V and T • if P is constant, gases expand when heated • when T increases, gas molecules move faster and collide with the walls more often and with greater force • to keep the P constant, the V must increase
Charles’ Law: V and T • Charles’ Law: the V of fixed mass of gas at constant P varies directly with Kelvin T. • V = kT • k is a constant for a certain sample of gas that depends on the mass of gas and P • What kind of graph is V vs. T?
Charles’ Law: V and T • discovered by French physicist, Jacques Charles in 1787 • first person to fill balloon with hydrogen gas and make solo balloon flight
Example: Charles’ Law & Temp. A sample of gas at 15°C and 1 atm has a volume of 2.58 L. What volume will this gas occupy at 38°C and 1 atm?
Gay-Lussac’s Law: P and T • Gay-Lussac’s Law: the P of fixed mass of gas at constant V varies directly with Kelvin T. • What kind of graph is P vs. T?
Example: Gay-Lussac’s Law The gas in an aerosol can is at a pressure of 3.00 atm at 25°C. Directions on the can warn the user not to keep the can in a place where temperature exceeds 52°C. What would the gas pressure be in the can at 52°C?
Way to remember which law goes with P,V and T Boyle’s P V T Gay-Lussac’s Charles’
Combined Gas Law • P1V1 = P2V2 T1 T2
A cylinder of a gas is kept at a constant volume, as the temperature increases from 24.1 C to 326.4 C. If the initial pressure was 1.10 atm, what is the final pressure in mm Hg?