Solids
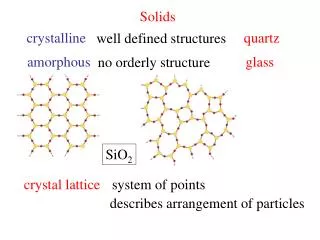
Solids. Solids have “resistance” to changes in both shape and volume Solids can be Crystalline or Amorphous Crystals are solids that consist of a periodic array of atoms, ions, or molecules

Solids
E N D
Presentation Transcript
Solids • Solids have “resistance” to changes in both shape and volume • Solids can be Crystalline or Amorphous • Crystals are solids that consist of a periodic array of atoms, ions, or molecules • If this periodicity is preserved over “large” (macroscopic) distances the solid has “Long-range Order” • Amorphous solids do not have Long-Range Order • Short Range Order
Crystals Solids: Short-range Order Long-range Order Amorphous solids: ~Short-range Order No Long-range Order Solids
Crystals • The periodic array of atoms, ions, or molecules that form the solids is called Crystal Structure • Crystal Structure = Space (Crystal) Lattice + Basis • Space (Crystal) Lattice is a regular periodic arrangement of points in space, and is purely mathematical abstraction • Crystal Structure is formed by “putting” the identical atoms (group of atoms) in the points of the space lattice • This group of atoms is the Basis
Crystals Crystal Structure = Space Lattice + Basis
Solids • Different solids can have the same geometrical arrangements of atoms • Properties are determined by crystal structure, i.e. both crystal lattice and basis are important • Example: • Si, Diamond (C), GaAs, ZnSe have the same geometry • Si and C (Diamond) Form “Diamond Structure” • GaAs or ZnSe form a structure called “Zinc Blende” http://www.neubert.net/Crystals/CRYStruc.html
Solids • Different arrangements of atoms (even the same atoms) give different properties Single layer is graphene
Solid Models: Close-Packed Spheres • Most atoms or ions forming solids have spherical symmetry • Considering the atoms or ions as solid spheres we can imagine crystals as closely packed spheres
Classification of Solids • Since we know the structure of atoms that form solids, we can classify them via the type of bonds that hold solids together • In this case we say that we classify solids according to the nature of bonding • There are four classes of solids: • metallic, ionic, covalent, and molecular • All the forces holding solids together have electrostatic origin
General Considerations • There must be an attractive force • An apparent candidate is the Coulomb Force • Here r is a distance between atoms (ions) forming a solid • What stops atoms (ions) from getting closer than they do? • When ions are very close to each other, other forces arise. These are the so-called short-range repulsive forces, due to rearrangement of electrons as nuclei approach • Equilibrium distance, r0, is point at which energy is at a minimum, forces are balanced
Ionic Solids • Ionic crystals consist of the negative and positive ions, attracted to each other • Electron from one of the atoms removed and transferred to another: NaCl, AgBr, KCl • When the crystal is formed excess heat is generated
Ionic Solids • Let’s find the energy required to transfer an electron from Na to Cl and then to form a NaCl molecule • To remove an electron from Na (ionize the atom) one needs to “spend” 5.14eV (compare with the ionization energy of a hydrogen atom?) Na + 5.14eV Na+ + e- • When a Cl atom captures an electron, 3.62eV of energy is released Cl + e- Cl- + 3.62eV
Ionic Solids • In solid, Na+ and Cl- are brought together at the distance r02.51Å [Å = 10-10m = 0.1nm] • The total energy is lowered due to the Coulomb attraction • The results is -5.73 eV • Thus when a NaCl “molecular unit” of NaCl solid is formed the following occurs Na + 5.14eV Na+ + e- Cl + e- Cl- + 3.62 eV Cl- + Na+ NaCl + 5.73 eV Na + Cl NaCl + 4.21 eV
Ionic Solids • The energy gain for NaCl solid is ~ 4.21 eV per NaCl pair • This is the energy required to break an NaCl molecule and restore neutral Na and Cl atoms • This energy is huge (in 1 cm3): 4.21×1.6×10-19 (Joules per pair) × 3 × 1022 (pairs) = 20200 Joules • To more accurately calculate the total electrostatic energy, need to calculate interaction of each ion with all other ions in the crystal
Ionic Solids • In ionic crystals all electrons are bound to the ions: There are no free electrons!!! • Thus most ionic crystals are insulators • There are ionic conductors, where ions, and not electrons conduct: Example: AgI
Covalent Solids • The covalent bond is usually formed from two electrons, one from each atom participating in the bonding: These electrons are shared by the atoms • Quantum Mechanics is required to calculate binding energies • The probability of finding electrons forming the bond between the two atoms is high • Covalent bonds are very strong and directional
Covalent Solids • In general, since there are no free electrons, these crystals are insulators or semiconductors
Compare • Covalent Solids • Ionic Solids
Mixed Bonding Solids • The electrons forming the covalent bond tend to be localized in the region between the two atoms • If the atoms elements forming the covalent solid are different • e.g., Zn & Se for ZnSe then the electrons a localized closer to one of the atoms (with higher electron affinity) • We say that the bond is partially covalent and partially ionic
Metallic Bond • Metals may be seen as collections of stationary ions surrounded by a sea of electrons • Can be viewed as limit of covalent bonding, when electrons are shared by all the ions in the crystal • The metallic bond is not directional
Molecular Solids • Very weak bonding: Dipole-Dipole Interaction • Liquid Crystals, Ice • low melting temperatures
Classification of Solids by Conductivity • Another way of classifying solids, in addition to (a) crystal structure and (b) type of bonds between atoms is (c) electrical conductivity • Conductors • Insulators • Semiconductors