CARBON
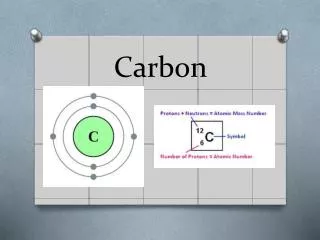
CARBON. ATOMIC NUMBER: 6 ATOMIC MASS: 12.01 ATOMIC SYMBOL: C CLASSIFICATION: NON-METALLIC COLOR: BLACK, COLORLESS. FUN, FAST FACTS. ATOMIC STRUCTURE. HISTORY OF CARBON. THIS ELEMENT OF PREHISTORIC DISCOVERY IS FOUND THROUGHOUT NATURE FOUND IN STARS, SUN, COMETS, ATOMSPHERES

CARBON
E N D
Presentation Transcript
ATOMIC NUMBER: 6 ATOMIC MASS: 12.01 ATOMIC SYMBOL: C CLASSIFICATION: NON-METALLIC COLOR: BLACK, COLORLESS FUN, FAST FACTS
HISTORY OF CARBON • THIS ELEMENT OF PREHISTORIC DISCOVERY IS FOUND THROUGHOUT NATURE • FOUND IN STARS, SUN, COMETS, ATOMSPHERES • THE ENERGY OF THE SUN AND THE STARS CAN BE ATTRIBUTED TO THE CARBON CYCLE
CARBON CAN BE FOUND IN THREE DIFFERENT FORMS: GRAPHITE DIAMONDS FULLERENE (aka BUCKYBALLS) 3 TYPES OF CARBON
GREAT GRAPHITE • LAYERED ATOMIC STRUCTURE-THERFORE PRODUCES A WEAK FORCE, AND IS EXTREMELY BREAKABLE • SOFT, SLIPPERY • MOST COMMONLY USED AS PENCIL LEAD
DIAMOND DATA • DIAMONDS HAVE AN ATOMIC SHAPE THAT ENSURES A TIGHT BOND • EACH CARBON ATOM IS THE SAME DISTANCE FROM ANOTHER ATOM • DIAMONDS ARE USED FOR CUTTING HARD OBJECTS AND FOR DECORATION (JEWELERY)
-ALSO KNOWN AS THE ‘BUCKYBALL” -RECENTLY DISCOVERED -CONSISTS OF 60 CARBON ATOMS -C60 -STILL BEING RESEARCHED FULLERENE
“BUCKYBALL” • THIS COMPLEX MOLECULE CAN BE ALTERED WITH A SINGLE ATOM TO CHANGE ITS PROPERTIES • NAMED AFTER ROBERT BUCKMINISTER FULLER • THIS MOLECULE HAS BEEN MAKING HEADLINES IN NANOTECHNOLOGY AND HOW THE DRUG INDUSTRY HAS INTEREST IN IT
COSTS OF CARBON -AS OF 1990, CARBON-13 WAS COMMERICIALLY AVAILABLE AT A COST OF ABOUT $700/GRAM.
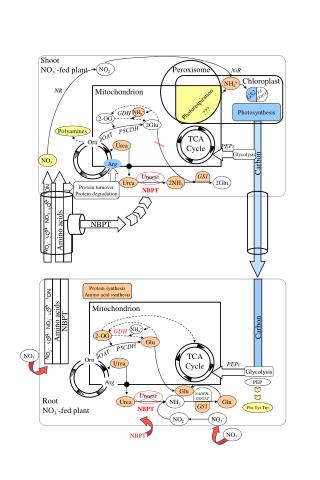
CARBON IN THE LITHOSPHERE • PLANTS ABSORB CARBON DIOXIDE FROM ATMOSPHERE, AND USE IT TO MAKE SUBSTANCES NEEDED FOR GROWTH • PHOTOSYNTHESIS INCORPORATES CARBON ATOMS FROM C02 TO SUGARS
LITHOSPHERE CONT. • ANIMALS (RABBIT) EAT PLANTS AND USE CARBON TO BUILD TISSUES • OTHER ANIMALS (FOX) EAT RABBIT AND USE CARBON FOR OWN NEEDS • ANIMALS RETURN C02 INTO AIR WHEN THEY BREATHE, AND WHEN THEY DIE (BECAUSE CARBON IS RETURED TO SOIL DURING DECOMPOSITION)
C02 ABSORBED INTO BODIES OF H20 SOME OF DISSOLVED C02 REMAINS IN WATER WHILE SOME IS USED BY ALGAE AND PHYTOPLANKTON THROUGH PHOTOSYNTHESIS MOLLUSCS (MUSSELS, CLAMS AND OYSTERS) MAKE SHELLS OF CALCIUM CARBONATE FROM C02 CARBON IN THE HYDROSPHERE
HYDROSPHERE CONTINUED • ONCE MUSSLES DIE, SHELLS FALL TO BOTTOM WITH HARD CALCIUM CARBONATE AND BECOMES COMPRESSED, CEMENTED TOGETHER, AND EVENTUALLY FOSSILISED INTO ROCKS LIKE CHALK, LIMESTONE, AND MARBLE
COMBUSTION OCCURS WHEN ORGANIC MATERIAL IS REACTED (BURNED) IN PRESENCE OF OXYGEN TO GIVE OFF C02 -DURING CONBUSTION, OXYGEN IS USED AND CABON DIOXIDE IS PRODUCED PLUS WATER CARBON IN THE ATMOSPHERE
SMOKESTACK • ORGANIC MATERIAL CAN BE ANY FOSSIL FUEL SUCH AS NATURAL GAS, (METHANE) OIL, OR COAL
PHOTOSYNTHESIS CARBON DIOXIDE+ WATER (h20) -> CARBOHYDRATE+ OXYGEN
RESPIRATION CARBOHYDRATE + OXYGEN-> CARBON DIOXIDE + OXYGEN
CALCIUM CARBONATE CALCIUM CARBONATE-> CALCIUM OXIDE + CARBON DIOXIDE
OIL AND COAL • WHEN TREES DIE, THEY FALL INTO SWAMPY MUD, WITH A LACK OF OXYGEN. THEREFORE, NORMAL DECOMPOSITION CANNOT OCCUR
OIL AND COAL II • TREES BECOME FOSSILISED (COAL IS THEN FORMED) • OIL FORMS FROM TINY DEAD PLANTS TRAPPED AT THE BOTTOM OF THE OCEAN OR MURKY WATER
RELATE COAL TO CARBON • THE PROCESS IN WHICH OIL AND COAL ARE MADE, IS RELATIVE TO THE CARBON CYCLE BECAUSE ALL LIVING ORGANISMS AND THE PROCESSES ARE MADE UP OF CARBON
RECAP OF CYCLES HYDROSPHERE, LITHOSPHERE, AND ATMOSPHERE AGAIN!
WHERE THE CYCLE BEGINS… • THE ATMOSPHERE CONTAINS CARBON DIOXIDE • THROUGH THE PROCESS OF PHOTOSYNTHESIS, PLANTS TAKE IN CARBON, CONTINUING THE CYCLE IN THE LITHOSPHERE • WHEN PHOTOSYNTHESIS EXCEEDS RESPIRATION, ORGANIC MATTER BUILDS UP TO FORM COAL AND OIL DEPOSITS
“The major exchange of carbon between the atmosphere and the lithosphere results from photosynthesis and respiration”
CARBON AND RESPIRATION • RESPIRATION IS PART OF THE PROCESS OF THE CARBON CYCLE IN THE LITHOSPHERE • ONCE ANIMALS BREAK DOWN CARBOHYDRATES IN PLANT TISSUES, THEY RELEASE IT TO THE ATMOSPHERE (THE CYCLE THEN CONINUES BACK TO THE LITHOSPHERE OR GOES TO THE HYDROSPHERE)
CARBON AND RESPIRATION CONTINUED • WHEN ANIMALS RELEASE CARBON DIOXIDE TO THE ATMOSPHERE, THEY DECOMPOSE, DECAY, AND FROM POOLS OF ENERGY CALLED FOSSIL FULES (INCLUDING COAL, OIL, AND NATURAL GASES)
CARBON IN THE HYDROSPHERE CARBON IS ABSORBED IN THE WATER BY PHYTOPLANKTON TO MAKE SHELL OF CALCIUM CARBONATE AND BECOME SEDIMENTS ONCE THEY DIE
CARBON IN HYDROSPHERE CONT. • CARBON DIOXIDE EXCHANGE IS CONTROLLED BY THE SEA SURFACE TEMPERATURE, CIRCULATING CURRENTS, AND THE BIOLOGICAL PROCESS OF PHYTOSYNTHESIS AND RESPIRATION
MORE CARBON IN HYDROSPHERE • CARBON DIOXIDE CAN DISSOVLE EASILY AND THE AMOUNT THAT THE OCEAN CAN HOLD DEPENDS ON THE OCEAN TEMPERATURE AND HOW MUCH CARBON IS ALREADY IN THE OCEAN • WHEN THE OCEAN IN COLD, MORE CARBON IS ARBORBED, AND WHEN THE TEMP. IS WARM, CO2 IS RELEASED TO THE SURFACE
THE GREENHOUSE EFFECT CARBON DIOXIDE CONTRIBUTES
KEEPS ALL EARHT’S HEAT FROM ESCAPING TO THE OUTER ATMOPSHERE CARBON RESPONSIBLE FOR GREENHOUSE GASES AND 55% OF THE GREENHOUSE EFFECT CARBON DIOXIDE IS TRANSPARENT TO LIGHT BUT OPAQUE TO HEAT RAYS, THEREFORE THE ATMOSPHERE RETARDS THE RAIATION OF HEAT FROM THE EARTH BACK INTO SPACE THE GREENHOUSE EFFECT
BIBLIOGRAPHY • PICTURE RESOURCES: • www.pmel.noaa.gov/ co2/gif/globcar.png • www.safeclimate.net/. ../carboncycle.php • geosun1.sjsu.edu/ ~dreed/105/exped7/4.html • www.cotf.edu/ete/modules/ carbon/efcarbon.html • www.webelements.com/.../ text/C/xtal.html • www.netl.doe.gov/.../ carbon-flux-diagram • www.mkdandj.com/ loosediamonds/
CONTINUED • 13 September 2004. http://library.thinkquest.org/11226/why.htm. • 13 September 2004. www.co2science.org/ • 13 September 2004. http://earthobservatory.nasa.gov/library/carboncycle • 13 September 2004. www.elmhurst.edu/~chm/vchembook/306carbon.html • 17 September 2004. www.nyu.edu/pages/mathmol/modules/carbon/carbon1.html • 17 September 2004. pearl1.lanl.gov/periodic/elements/6.html
EVEN MORE!!!!!!! • 17 September 2004. www.environment.allinfoabout.com/carbon.html • 19 September 2004. www.cotf.edu/ete/modules/climate/gccarbon1.html