CARBON
CARBON. By: Tristan Bowman. CARBON. Atomic Symbol – C Atomic Number – 6 Atomic Mass – 12.011 Group Number – 14 Period Number - 2. Atom of Carbon. PHYSICAL PROPERTIES.

CARBON
E N D
Presentation Transcript
CARBON By: Tristan Bowman
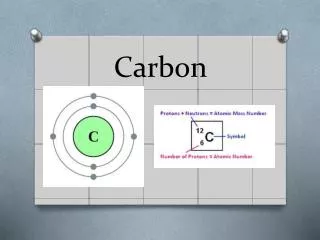
CARBON Atomic Symbol – C Atomic Number – 6 Atomic Mass – 12.011 Group Number – 14 Period Number - 2
PHYSICAL PROPERTIES Melting Point – 3500° C (6400 ° F) Boiling Point – 4027° C (7281° F) Liquid Range – 500 K Density of Solid – 2267 Kg m-3 Molar Volume – 5.29cm³ Velocity of Sound – 18350 m s-1
Chemical Properties Carbon does not dissolve in or react with water, acids, or most other materials. One thing Carbon does react with is Oxygen, which makes (CO2)- Carbon Dioxide and (CO)- Carbon Monoxide.
USES OF CABON Carbon is used to make: Graphite, Diamonds, Coal, Charcoal, the black color in inks, pigments(paints), rubber tires, stove polish, typewriter ribbons, and Phonograph records
HISTORY OF CARBON Carbon has been around since before 509 B.C. René Antoine Ferchault Reaumur was one of the first French Physicists to believe that Carbon was an element Carbon was officially classified as an element near the end of the eighteenth century
ALLOTROPES Diamonds Graphite Graphene Amorphous Fullerene ISOTOPES Carbon- 12 Carbon- 13 Carbon-14
IMPORTANT COMPOUNDS Carbon Dioxide(CO2) Carbon Monoxide(CO) Carbon Disulfide(CS2) Chloroform(CHCL3) Carbon Tetrachloride(CCL4) Methane(CH4) Ethylene(C2H4) Acetylene(C2H2) Benzene(C6H6) Ethyl Alcohol(CH5OH) Acetic Acid(CH3COOH)
INTERESTING FACTS Carbon only has two stable Isotopes. Carbon is the sixth most common element in the universe and the fourth most common element in the solar system. Carbon is the second most common element in the human body after Oxygen. About 18 percent of a person’s body weight is due to Carbon.