Carbon
Carbon. 2-3 Bio. You will need a textbook & your notebook. Carbon. http:// www.chemicalelements.com/elements/c.html. Organic Chemistry. Study of all compounds that contain bonds between carbon atoms. Chemistry of Carbon. Carbon atoms have 4 valence electrons

Carbon
E N D
Presentation Transcript
Carbon 2-3 Bio You will need a textbook & your notebook
Carbon http://www.chemicalelements.com/elements/c.html
Organic Chemistry • Study of all compounds that contain bonds between carbon atoms.
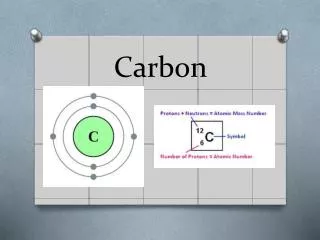
Chemistry of Carbon • Carbon atoms have 4 valence electrons • Each electron can join with an electron from another atom to form a strong covalent bond • C can bond with many elements including: • H • O • P • S • N
Chemistry of Carbon • Most importantly Carbon can bond to another Carbon • It can form single, double, or triple bonds • These bonds can create long chains or even ring structures • Millions of different large and complex structures can be formed • No other element even come close to matching carbons versatility
Draw the following in your notebook: • Methane • What elements? What types of bonds? • Acetylene • What elements? What types of bonds? • Benzene • What elements? What types of bonds?
Macromolecules • Biomolecules • Large molecules found in living cells
Polymerization • Process by which macromolecules are formed • Small units (monomers) link together to form long chains (polymers)
Monomers • Example: glucose
Dehydration synthesis • Process of removing water to create a bond that connects two smaller molecules (monomers) to form larger molecules (polymers) • To form the water molecule that is removed, one monomer givers –OH the other gives -H
Hydrolysis • Breaking chemical bond between to monomers by inserting a water molecule. • One atom gets an OH group the other gets a H
Write equations for Dehydration synthesis R-OH + HO-R = R-O-R + H20 IS DEHYDRATION Hydrolysis R-O-R= R-OH + HO-R IS HYDROLYSIS.
Demonstrate: • The formation of a monomer • Destruction of a polymer
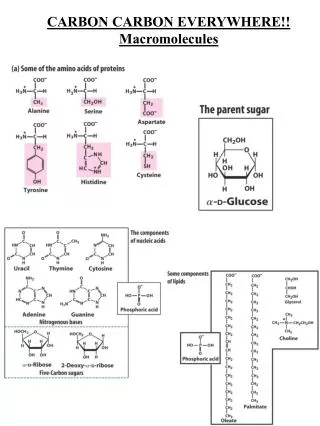
Macromolecules • 4 classes/groups • Proteins • Carbohydrates • Lipids • Nucleic Acids
Day 2 Notes • You will need your notebook and a textbook
Carbohydrates • Contain C, H, & O • In a 1:2:1 Ratio • Example Glucose: • Chemical Formula: • C6H12O6
Carbohydrates- function: • Living things use carbohydrates as their main source of energy. • Plants and some animals use carbohydrates for structural purposes
Carbohydrate Terminology: • Monosaccharide- simple sugar • Disaccharides- two sugars joined • Polysaccharides- more than 2 joined
Monosaccharides • Glucose • Galactose • fructose
Disaccharides • Sucrose • Maltose • Lactose http://www.biocab.org/Disaccharides.html
Polysaccharides • Starch- in plants-long chains of glucose • Glycogen-stored in animal liver • Cellulose- plants-wood & fiber
Testing for carbohydrates • Simple Sugar (monosaccharide) test: • Indicator: • positive: • Negative • Starch test (polysaccharide/ complex carb) test • Indicator: • Positive: • Negative:
Structure: • http://www.chemicalformula.org/sugar
Day 3 Notes: • Lipids
Lipids • Large & varied group of biomolecules • Generally not soluble in water
Lipids: Made mostly of Carbon & Hydrogen atoms http://biology.clc.uc.edu/courses/bio104/lipids.htm
Triglycerides • 3 fatty acids are attached
Glycerol Functional groups?
Common categories of Lipids: • Fats • Oils • Waxes
Functions / Uses: • Energy storage • Important parts of biological membranes • Waterproof coverings • Chemical messengers
Chemical messengers • Steroids Cholesterol • http://biology.clc.uc.edu/courses/bio104/lipids.htmChosterol
Cholesterol The general structure of cholesterol consists of two six-membered rings side-by-side and sharing one side in common, a third six-membered ring off the top corner of the right ring, and a five-membered ring attached to the right side of that. The central core of this molecule, consisting of four fused rings, is shared by all steroids, including estrogen (estradiol), progesterone, corticosteroids such as cortisol (cortisone), aldosterone, testosterone, and Vitamin D. In the various types of steroids, various other groups/molecules are attached around the edges. Know how to draw the four rings that make up the central structure.
Lipid formation: • Glycerol molecule combines with fatty acids • Figure 2-14 page 46
Saturated vs. Unsaturated Fats • Saturated = max # of Hydrogen atoms on each Carbon of the Fatty acid (saturated with H) • Unsaturated= at least one carbon-carbon double bond in the fatty acid • Polyunsaturated = ___________
Common unsaturated Fats • One double bond = Monounsaturated • Liquid at room temperature: • Olive oil
Common Polyunsaturated Fats (Many Common Cooking Oils) • Corn oil • Sesame oil • Canola oil • Peanut oil
Meat Products • Contain both saturated & unsaturated • FDA recommends
Saturated fats: • Popular among manufacturers of processed foods because • they are less vulnerable to rancidity • More solid at room temperature • Higher melting point
Unsaturated fats • Lower melting point so more fluidity of cell membranes • Replacing Saturated fats w/unsaturated fats helps to lower levels of total cholesterol & LDL cholesterol in the blood. • Foods: avacado, nuts, veg oil • Although the better fat, FDA Recommends Diet with < 30% of calories (67g in 2000 Cal diet)
Trans unsaturated fats: • Describe the attachment of the H to the C =C bond • Should be avoided = high health risks--- plaque in arteries--- coronary atherosclerosis
Lipid test Indicator: Sudan IV test Negative: Positive: