Subatomic Particles
Subatomic Particles. Electrons, Protons and Neutrons and Their Electrical Charge. MATTER. All objects contain matter All matter contains tiny particles in constant motion All matter is made from different proportions of a small number of the basic building blocks….Elements

Subatomic Particles
E N D
Presentation Transcript
Subatomic Particles Electrons, Protons and Neutrons and Their Electrical Charge
MATTER • All objects contain matter • All matter contains tiny particles in constant motion • All matter is made from different proportions of a small number of the basic building blocks….Elements • WHAT IS AN ELEMENT? • A pure substance that cannot be broken into a simpler form
ATOMS: • Atoms are the smallest particles that make up matter • Atoms are the smallest part of an element that retains the properties of that element What is a Compound? A substance that contains 2 or more different elements joined together by chemical bonds
ATOMIC THEORY • Dalton’s Theory from 1808 1. Matter is composed of tiny, invisible, indestructible particles called atoms 2. An element is composed entirely of one type of atom. The properties of all atoms of one particular element are identical. 3. A compound contains atoms of 2 or more different elements. 4. Atoms do not change their identities in chemical reactions, they are just rearranged.
EXCEPTIONS TO DALTON’S THEORY • We now know that atoms are not indivisible, but are made of smaller particles. • Atoms are not indestructible, but can be split. • Not all of the atoms of an element are exactly identical.
Subatomic Particles • The Atom is comprised of 3 subatomic particles • These particles are known as the… • Proton (+) • Neutron (+/-) • Electron (-)
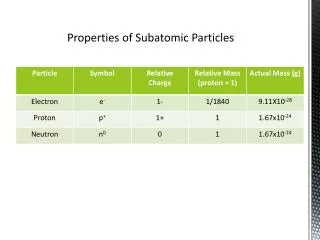
Subatomic Particles There are 3 subatomic particles that make up an atom… 1. The Proton: A positively charged particle that resides in the nucleus. 2. The Neutron: A neutral particle that resides in the nucleus along with the proton, and holds the nucleus together 3. The Electron: A negatively charged particle that circles the central part of the atom in Orbital Rings The charges of the proton and electron are equal and opposite
Mass and Volume of an Atom The protons and neutrons within the nucleus are responsible for giving the atom its mass • The electrons have little to no mass The distance between the electrons and the nucleus is what gives the atom its volume
Atomic Number/Atomic Mass • The number of protons in an atom’s nucleus give it its atomic number. • The mass of the protons + the mass of the neutrons are what give an atom its atomic mass • These are numbers that help identify elements on the Periodic Table of Elements
STATIC ELECTRICITY A charge is a fundamental property due to subatomic particles Static electricity is electricity that is created and remains stationary It is a charge that is created by friction or the rubbing of surfaces
Van DeGraaff Generator WHAT IT CAN DO HOW IT WORKS
STATIC CHARGES • Objects can become charged • Protons are bound to the nucleus, but electrons can easily be stripped from an object • Objects receiving electrons becomes negatively charged • Objects losing electrons become positively charged
DEMONSTRATION • Static Demonstration • Another Static Demonstration • When you bring a negatively charged item near a neutral object, you get a static discharge. • The spark and snap is the ELECTRONS jumping creating a glow
FIELDS AND FORCES • A FIELD is how the force of electricity, light or gravity gets from one place to another • All interaction between matter and energy will proceed because of the fields • We are experiencing the fields created by an object NOT the object itself
INVERSE SQUARE LAW • The strength of a field will decrease as the distance between objects increases. • The relationship is negative and is calculated as the inverse square of the distance. • Example: If you increase the distance/double the distance between you and a light source, you decrease the field strength to ¼ of its original strength
ELECTRIC FIELDS: • Fields are indicated by drawing “FIELD LINES” • For electric fields, the field lines are moving away from a + and towards a _