Calculating Average Atomic Mass Using Isotope Abundances
This guide explains how to calculate the average atomic mass of an element using the relative abundances of its isotopes. Since actual atomic masses are so small, they are expressed in atomic mass units (amu), with carbon-12 assigned a mass of exactly 12 amu. To find the average atomic mass, convert isotope abundance percentages to decimals, multiply these by their respective atomic masses, and sum the products. For example, when calculating chlorine's atomic mass using its isotopes with masses of 34.969 amu (75.77% abundance) and 36.966 amu (24.33% abundance), the average atomic mass is found to be approximately 35.48 amu.

Calculating Average Atomic Mass Using Isotope Abundances
E N D
Presentation Transcript
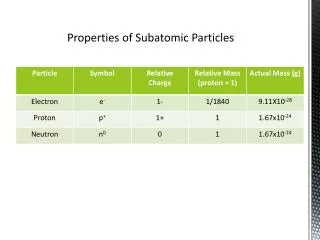
Calculating Average Atomic Mass • expressed in atomic mass units (amu) • done because actual masses so small and hard to work with • based on carbon-12 assigned mass of exactly 12 amu • weighted average of all naturally occurring isotopes.
change %abundances to decimals for each isotope Multiply abundance by atomic mass of isotope Add products to get average atomic mass for element for 75.77% = 0.7577 for 24.33% = 0.2433 Example Chlorine has two isotopes. The isotope with a mass of 34.969 amu has a relative abundance of 75.77%. The isotope with a mass of 36.966 amu has a relative abundance of24.33%. Calculate the atomic mass of chlorine. x 34.969 = 26.49 amu x 36.966 = 8.994 amu for Cl atomic mass = 35.484 = 35.48 amu