Notes on Subatomic Particles
Notes on Subatomic Particles. proton. neutron. There are 3 subatomic particles in an atom: 1. 2. 3. electron. nucleus. Electrons. (negative). Proton Neutron. (positive). (neutral). 1 amu (atomic mass unit). positive. nucleus. (p + ). neutral. nucleus. 1 amu. (n).

Notes on Subatomic Particles
E N D
Presentation Transcript
Notes on Subatomic Particles proton neutron There are 3 subatomic particles in an atom: 1. 2. 3. electron
nucleus Electrons (negative) Proton Neutron (positive) (neutral)
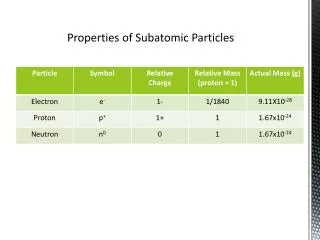
1 amu (atomic mass unit) positive nucleus (p+) neutral nucleus 1 amu (n) Zip around nucleus in shells (e-) negative _1_ amu 1837
When using the periodic table to find subatomic particle information… Atomic mass # symbol A Z Atomic # Atomic mass # : • Is a number in decimal form • When you round it, it tells you the mass of p+ + n + e- x Atomic # : • Tells the # of p+ • Tells the # of e- To find the # of neutrons: Atomic mass # - atomic # = # neutrons
Atoms that aren’t typical • ISOTOPE – atom with a number of neutrons that is higher or lower than what’s indicated on the periodic table (typically radioactive) • ION – atom with a number of electrons that is higher (anion) or lower (cation) than what’s indicated on the periodic table
Examples on Subatomic Particle Worksheet: 23 sodium 11 11 12 11 11 91 Zr zirconium 40 40 40 40 Cs cesium 55 55 78 133 55
Notes on the Periodic Table • Periodic table created by Dimitri Mendeleev He organized it by increasing mass (doesn’t work…look at #27 and #28) and by similar properties • NOW organized by increasing atomic number and by similar properties
Organization of the Periodic Table Rows () – periods, shells, energy levels, electron clouds Columns (↕) – groups, families, # of valence electrons Left of Zig Zag Line - metals, cations (positive ions) Zig Zag Line – metalloids, semi-metals, semiconductors Right of ZigZag Line – non-metals, anions (negative ions)
Families of the Periodic Table • Column 1 – Alkali Metals • Column 2 – Alkaline Earth Metals • Column 8 (18) – Nobel Gases • Column 7 (17) – Halogens • Column 6 (16) – Oxygen Family * Column 5 (15) – Nitrogen Family **Note: Only Down to Zig Zag line for Oxygen Family and Nitrogen Family!!
Families of the Periodic Table continued… Middle of the periodic table … TRANSITION METALS • Coinage Family (Cu, Ag, Au) • Lanthanides (1st row on bottom) • Actinides (2nd row on bottom)