Subatomic Particles
Subatomic Particles. By Joshua Cohon. Nuclei. The nucleus lies at the center of the atom. It contains the protons and neutrons. It contains positive charge and no charge, so it is a positive charge. Plural for nucleus is nuclei. Protons. Protons have a positive charge.

Subatomic Particles
E N D
Presentation Transcript
Subatomic Particles By Joshua Cohon
Nuclei • The nucleus lies at the center of the atom. • It contains the protons and neutrons. • It contains positive charge and no charge, so it is a positive charge. • Plural for nucleus is nuclei.
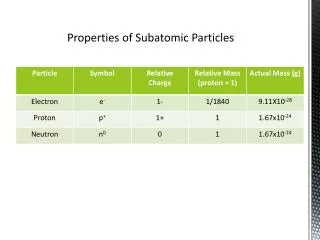
Protons • Protons have a positive charge. • The atomic number of an element tells how many protons there are in one of its atoms. • The element that has the most protons is unniloctium at 118 protons.
Neutrons • Neutron are neutral. • That means that they have no charge. • Sometimes, electrons and protons form together to make neutrons.
Electrons • Electrons orbit the nuclei. • Electricity is formed by electrons flowing. • Sometimes, they are found outside of atoms.
Quarks • Quarks build up subatomic particles. • There are three of them in all nuclei- contained particles. • In pions, there are two of them.
Fun Facts • When you see anti in front of the name of a subatomic particle, it is antimatter, the complete opposite of matter! • 1 million atoms stretch 1 inch!
Questions • How many quarks are in a proton? • Is a pion nuclei-contained? • What color quarks are there?