II. Crystal Structure
II. Crystal Structure. Lattice, Basis, and the Unit Cell Common Crystal Structures Miller Indices for Crystal Directions and Planes The Reciprocal Lattice.

II. Crystal Structure
E N D
Presentation Transcript
II. Crystal Structure • Lattice, Basis, and the Unit Cell • Common Crystal Structures • Miller Indices for Crystal Directions and Planes • The Reciprocal Lattice
An ideal crystalline solid is an infinite repetition of identical structural units in space. The repeated unit may be a single atom or a group of atoms. A. Lattice, Basis, and Unit Cell An important concept: crystal structure = lattice + basis = +
lattice: a periodic array of points in space. The environment surrounding each lattice point is identical. basis: the atom or group of atoms “attached” to each lattice point in order generate the crystal structure. • The translational symmetry of a lattice is given by the base vectors or lattice vectors . Usually these vectors are chosen either: • to be the shortest possible vectors, or • to correspond to a high symmetry unit cell
Example: a 2-D lattice These two choices of lattice vectors illustrate two types of unit cells: Conventional (crystallographic) unit cell: larger than primitive cell; chosen to display high symmetry unit cell Primitive unit cell: has minimum volume and contains only one lattice point
A lattice translation vector connects two points in the lattice that have identical symmetry: In our 2-D lattice:
B. Common Crystal Structures 2-D only 5 distinct point lattices that can fill all space 3-D only 14 distinct point lattices (Bravais lattices) The 14 Bravais lattices can be subdivided into 7 different “crystal classes”, based on our choice of conventional unit cells (see text, handout). Attaching a basis of atoms to each lattice point introduces new types of symmetry (reflection, rotation, inversion, etc.) based on the arrangement of the basis atoms. When each of these “point groups” is combined with the 14 possible Bravais lattices, there are a total of 230 different possible “space groups” in 3-D. We will focus on the few that are common for metals, semiconductors, and simple compounds.
Crystal Structure Diagrams (a) NaCl(b) CsCl (c) fluorite(d) perovskite (e) Laves phase(f) A15
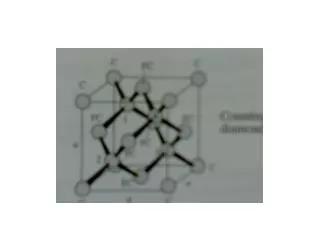
Crystal Structure Diagrams (continued) hexagonal close packed (Be, Mg, Zn) diamond structure (C, Si, Ge)
Analysis of Common Crystal Structures 1. NaCl structure (many ionic solids) lattice: face-centered cubic (fcc) basis: Na at 000, Cl at ½½½ 2. CsCl structure (some ionic solids and intermetallic alloys) lattice: simple cubic (sc) basis: Cs at 000, Cl at ½½½
Common Crystal Structures, cont’d 3. hexagonal-close-packed (divalent metals) lattice: hexagonal basis: 000, 2/3 1/3 1/2 (see text for an alternate choice of lattice and basis) 4. diamond structure (C, Si, Ge) lattice: face-centered-cubic (fcc) basis: 000, ¼¼¼ 5. zincblende structure (ZnS, GaAs, InP, compound semicond’s) lattice: face-centered-cubic (fcc) basis: Zn at 000, S at ¼¼¼
z z = 3 y = 2 y x = 1 x C. Miller Indices for Crystal Directions & Planes Because crystals are usually anisotropic (their properties differ along different directions) it is useful to regard a crystalline solid as a collection of parallel planes of atoms. Crystallographers and CM physicists use a shorthand notation (Miller indices) to refer to such planes. 1. Determine intercepts (x, y, z) of the plane with the coordinate axes
2. Express the intercepts as multiples of the base vectors of the lattice C. Miller Indices,cont’d. In this example, let’s assume that the lattice is given by: Then the intercept ratios become: 3. Form reciprocals: 4. Multiply through by the factor that allows you to express these indices as the lowest triplet of integers: We call this the (212) plane.
z Another example Find the Miller indices of the shaded plane in this simple cubic lattice: a y a a x Intercepts: non-intersecting intercept at Intercept ratios: Reciprocals: We call this the (010) plane. Note: (hkl) = a single plane; {hkl} = a family of symmetry-equivalent planes
z y x Crystal Planes and Directions Crystal directions are specified [hkl] as the coordinates of the lattice point closest to the origin along the desired direction: Note: [hkl] = a specific direction; <hkl> = a family of symmetry-equivalent directions Note that for cubic lattices, the direction [hkl] is perpendicular to the (hkl) plane
Crystal planes (hkl) in the real-space or direct lattice are characterized by the normal vector and the interplanar spacing : D. The Reciprocal Lattice z y x Long practice has shown CM physicists the usefulness of defining a different lattice in reciprocal space whose points lie at positions given by the vectors This vector is parallel to the [hkl] direction but has magnitude 2/dhkl, which is a reciprocal distance
The reciprocal lattice is composed of all points lying at positions from the origin, so that there is one point in the reciprocal lattice for each set of planes (hkl) in the real-space lattice. The Reciprocal Lattice, cont’d. This seems like an unnecessary abstraction. What is the payoff for defining such a reciprocal lattice? • The reciprocal lattice simplifies the interpretation of x-ray diffraction from crystals (coming soon in chapter 3) • The reciprocal lattice facilitates the calculation of wave propagation in crystals (lattice vibrations, electron waves, etc.)
The Reciprocal Lattice: An Analogy In the analysis of electrical signals that are periodic in time, we use Fourier analysis to express such a signal in the frequency domain: If f(t) has period T, then the coefficient C is nonzero only for frequencies given by n = integer Waves of lattice vibrations or electron waves moving through a crystal with a periodicity specified by base vectors can likewise be decomposed into a sum of plane waves: Here, the coefficient Ck is nonzero only when the vector k is a reciprocal lattice translation vector: A, B, and C are the base vectors of the reciprocal lattice (some books use a*, b*, c*)
Definition of Reciprocal Lattice Base Vectors These reciprocal lattice base vectors are defined: Which have the simple dot products with the direct-space lattice vectors: So compare, for example: frequency time Reciprocal lattice direct lattice
Remember: Problems worthy of attack Prove their worth by hitting back --Piet Hein