CRYSTAL STRUCTURE
CRYSTAL STRUCTURE. Crystal Structure. Objectives Relationships between structures-bonding-properties of engineering materials. Arrangements in crystalline solids Give examples of each: Lattice, Crystal Structure, Unit Cell and Coordination Numbers

CRYSTAL STRUCTURE
E N D
Presentation Transcript
Crystal Structure Objectives • Relationships between structures-bonding-properties of engineering materials. • Arrangements in crystalline solids • Give examples of each: Lattice, Crystal Structure, Unit Cell and Coordination Numbers • Describe hard-sphere packing and identify cell symmetry. • Define directions and planes (Miller indices) for crystals Outline • Structure of the Atom and Atomic Bonding • Electronic Structure of the Atom • Lattice, Unit Cells, Basis, and Crystal Structures • Points, Directions, and Planes in the Unit Cell • Crystal Structures of Ionic Materials • Covalent Structures
Crystal Structure Crystal Structure • Lattice- A collection of points that divide space into smaller equally sized segments. • Unit cell - A subdivision of the lattice that still retains the overall characteristics of the entire lattice. • Atomic radius - The apparent radius of an atom, typically calculated from the dimensions of the unit cell, using close-packed directions (depends upon coordination number). • Packing factor - The fraction of space in a unit cell occupied by atoms. Types of Crystal Structure • Body centered cubic (BCC) • Face centered cubic (FCC) • Hexagonal close packed (HCP)
Crystal Structure A number of metals are shown below with their room temperature crystal structure indicated. There are substances without crystalline structure at room temperature; for example, glass and silicone. All metals and alloys are crystalline solids, and most metals assume one of three different lattice, or crystalline, structures as they form: body-centered cubic (BCC), face-centered cubic (FCC), or hexagonal close-packed (HCP).
Number of Lattice Points in Cubic Crystal Systems Crystal Structure • In the SC unit cell: point / unit cell = (8 corners)1/8 = 1 • In BCC unit cells: point / unit cell = (8 corners)1/8 + (1 center)(1) = 2 • In FCC unit cells: point / unit cell = (8 corners)1/8 + (6 faces)(1/2) = 4 Relationship between Atomic Radius and Lattice Parameters In SC, BCC, and FCC structures when one atom is located at each lattice point.
Crystal Structure Packing Factor • In a FCC cell, there are four lattice points per cell; if there is one atom per lattice point, there are also four atoms per cell. The volume of one atom is 4πr3/3 and the volume of the unit cell is a0 3
Crystal Structure Density Density of BCC iron, which has a lattice parameter of 0.2866 nm. • Atoms/cell = 2, a0 = 0.2866 nm = 2.866 10-8 cm • Atomic mass = 55.847 g/mol • Volume of unit cell = = (2.866 10-8 cm)3 = 23.54 10-24 cm3/cell • Avogadro’s number NA = 6.02 1023 atoms/mol
Crystal Structure Points, Directions, and Planes in the Unit Cell Geometry Unit Cell: The basic structural unit of a crystal structure. A unit cell is the smallest component of the crystal that reproduces the whole crystal when stacked together with purely translational repetition. R R R R a Atomic configuration in Face-Centered-Cubic Atomic configuration in Simple Cubic
Crystal Structure Bravais Lattices
Crystal Structure Unit Cells Types A unit cell is the smallest component of the crystal that reproduces the whole crystal when stacked together with purely translational repetition. • Primitive (P) unit cells contain only a single lattice point. • Internal (I) unit cell contains an atom in the body center. • Face (F) unit cell contains atoms in the all faces of the planes composing the cell. • Centered (C) unit cell contains atoms centered on the sides of the unit cell. Face-Centered Primitive Body-Centered End-Centered Crystal Classes (cubic, tetragonal, orthorhombic, hexagonal, monclinic, triclinic, trigonal) with 4 unit cell types (P, I, F, C) symmetry allows for only 14 types of 3-D lattice.
Crystal Structure Counting Number of Atoms Per Unit Cell • Counting Atoms in 3D Cells • Atoms in different positions are shared by differing numbers of unit cells. • • Vertex atom shared by 8 cells => 1/8 atom per cell. • • Edge atom shared by 4 cells => 1/4 atom per cell. • • Face atom shared by 2 cells => 1/2 atom per cell. • • Body unique to 1 cell => 1 atom per cell. Simple Cubic • 8 atoms but shared by 8 unit cells. So, • 8 atoms/8 cells = 1 atom/unit cell How many atoms/cell for Body-Centered Cubic? And, Face-Centered Cubic?
= 0.74 Crystal Structure Atomic Packing Fraction for FCC Face-Centered-Cubic Arrangement APF = vol. of atomic spheres in unit cell total unit cell vol. No. of atoms per unit cell = Volume of one atom= Volume of cubic cell = “R” related to “a” by 4/cell 4R3/3 a3
Crystal Structure APF for BCC Again, For BCC • BCC: a = b = c = a and angles a = b =g= 90°. • 2 atoms in the cubic cell: (0, 0, 0) and (1/2, 1/2, 1/2).
A B C Crystal Structure FCC Stacking Highlighting the stacking Highlighting the faces
Crystal Structure FCC Stacking • ABCABC.... repeat along <111> direction gives Cubic Close-Packing (CCP) • Face-Centered-Cubic (FCC) is the most efficient packing of hard-spheres of any lattice. • Unit cell showing the full symmetry of the FCC arrangement : a = b =c, angles all 90° • 4 atoms in the unit cell: (0, 0, 0) (0, 1/2, 1/2) (1/2, 0, 1/2) (1/2, 1/2, 0)
A Crystal Structure HCP Stacking Highlighting the stacking B A Highlighting the cell Layer A Layer B Layer A
Crystal Structure HCP Stacking • ABABAB.... repeat along <111> direction gives Hexagonal Close-Packing (HCP) • Unit cell showing the full symmetry of the HCP arrangement is hexagonal • Hexagonal: a = b, c = 1.633a and angles a = b = 90°,g = 120° • 2 atoms in the smallest cell: (0, 0, 0) and (2/3, 1/3, 1/2).
pt. Crystal Structure Crystallographic Points, Directions, and Planes. To define a point within a unit cell…. Express the coordinates uvw as fractions of unit cell vectors a, b, and c (so that the axes x, y, and z do not have to be orthogonal). pt. coord. x (a) y (b) z (c) 0 0 0 origin 1 0 0 1 1 1 1/2 0 1/2
Crystal Structure Crystallographic Points, Directions, and Planes.
c b a • 5. Designate negative numbers by a bar • Pronounced “bar 1”, “bar 1”, “zero” direction. 6. “Family” of [110] directions is designated as <110>. DIRECTIONS will help define PLANES (Miller Indices or plane normal). Crystal Structure Crystallographic Points, Directions, and Planes. • Procedure: • Any line (or vector direction) is specified by 2 points. • The first point is, typically, at the origin (000). • Determine length of vector projection in each of 3 axes in units (or fractions) of a, b, and c. • X (a), Y(b), Z(c) • 1 1 0 • Multiply or divide by a common factor to reduce the lengths to the smallest integer values, u v w. • Enclose in square brackets: [u v w]: [110] direction.
Crystal Structure Crystallographic Points, Directions, and Planes.
Crystal Structure Miller Indices for HCP Planes 4-index notation is more important for planes in HCP, in order to distinguish similar planes rotated by 120o. t As soon as you see [1100], you will know that it is HCP, and not [110] cubic! Find Miller Indices for HCP: Find the intercepts, r and s, of the plane with any two of the basal plane axes (a1, a2, or a3), as well as the intercept, t, with the c axes. Get reciprocals 1/r, 1/s, and 1/t. Convert reciprocals to smallest integers in same ratios. Get h, k, i , l via relation i = - (h+k), where h is associated with a1, k with a3, i with a2, and l with c. Enclose 4-indices in parenthesis: (h k i l) . r s
The plane’s intercept a1, a3 and c at r=1, s=1 and t= , respectively. The reciprocals are 1/r = 1, 1/s = 1, and 1/t = 0. They are already smallest integers. We can write (h k i l) = (1 ? 1 0). Using i = - (h+k) relation, k=–2. Miller Index is Crystal Structure Miller Indices for HCP Planes What is the Miller Index of the pink plane?
Number of atoms centered on a direction vector LD = Length of the direction vector Crystal Structure Linear Density in FCC Example: Calculate the linear density of an FCC crystal along [1 1 0]. ASK a. How many spheres along blue line? b. What is length of blue line? ANSWER 2 atoms along [1 1 0] in the cube. Length = 4R XZ = 1i + 1j + 0k = [110]
Area of atoms centered on a given plane PD = Area of the plane Crystal Structure Planar Packing Density in FCC Example: Calculate the PPD on (1 1 0) plane of an FCC crystal. • Find area filled by atoms in plane: 2R2 • Find Area of Plane: 8√2 R2 Hence, Always independent of R!
Theoretical Density, Crystal Structure • crystal structure = FCC: 4 atoms/unit cell • atomic weight = 63.55 g/mol (1 amu = 1 g/mol) • atomic radius R = 0.128 nm
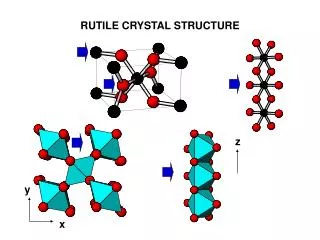
Crystal Structures of Ionic Materials Crystal Structure • Factors need to be considered in order to understand crystal structures of ionically bonded solids are: • Ionic Radii • Electrical Neutrality • Connection between Anion Polyhedra • Visualization of Crystal Structures Using Computers The cesium chloride structure, a SC unit cell with two ions (Cs+ and CI-) per lattice point. (b) The sodium chloride structure, a FCC unit cell with two ions (Na+ + CI-) per lattice point
Crystal Structures of Ionic Materials Crystal Structure Fluorite unit cell, (b) plan view. The zinc blende unit cell, (b) plan view.
Crystal Structures of Ionic Materials Crystal Structure The perovskite unit cell showing the A and B site cations and oxygen ions occupying the face-center positions of the unit cell.
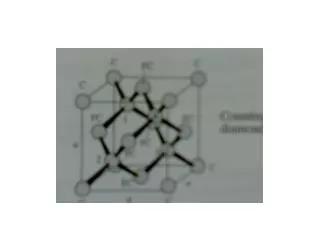
Covalent Structures Crystal Structure Tetrahedron and (b) the diamond cubic (DC) unit cell. This open structure is produced because of the requirements of covalent bonding The silicon-oxygen tetrahedron and the resultant β-cristobalite form of silica
Covalent Structures Crystal Structure Tetrahedron and (b) the diamond cubic (DC) unit cell. This open structure is produced because of the requirements of covalent bonding