Lecture 2: Crystal Structure
Lecture 2: Crystal Structure. PHYS 430/603 material Laszlo Takacs UMBC Department of Physics. Unless we specify otherwise, “solid” means “crystalline,” at least on the microscopic scale.

Lecture 2: Crystal Structure
E N D
Presentation Transcript
Lecture 2: Crystal Structure PHYS 430/603 material Laszlo Takacs UMBC Department of Physics
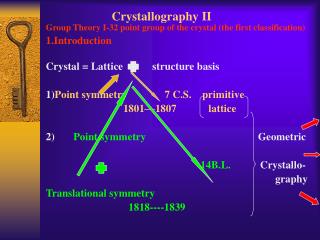
Unless we specify otherwise, “solid” means “crystalline,” at least on the microscopic scale • Short range structure reflects the nature of bonds, but the crystal structure also has to conform to translational symmetry: • If we shift the crystal by certain vectors of translation, T, every atom moves into the position of an identical atom. • The possible vectors of translation are linear combinations with integer coefficients of three “primitive translational vectors”: T = na + mb +pc • The entire structure can be described by a “unit cell” defined as a parallelepiped defined by a, b, c and its repeated translations by a, b, c. There can be symmetries beyond translation. • A smallest possible unit cell is the “primitive cell.” • The points in a lattice are mathematical points, we get the crystal structure by putting identical groups of atoms - the basis - on each lattice point. In simple cases, the basis is a single atom.
The elementary vectors of translation, i.e. the unit vectors of our coordinate system
Find the unit cell Maurits Cornelis Escher