Metallic bonding and properties
470 likes | 1.73k Views
Metallic bonding and properties. What properties do metals have?. Metallic bonding. Metallic bonding. The atoms are held together as ions with free electrons. The free electrons move through the outer shells with ease.

Metallic bonding and properties
E N D
Presentation Transcript
Metallic bonding and properties What properties do metals have?
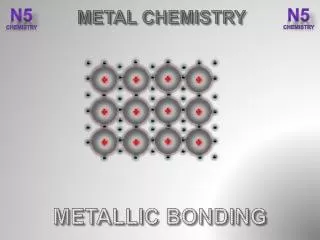
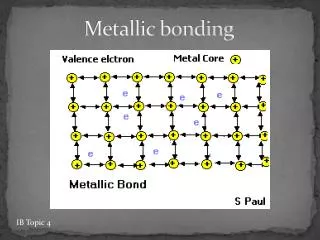
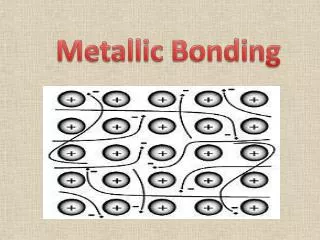
Metallic bonding • The atoms are held together as ions with free electrons. • The free electrons move through the outer shells with ease. • This ‘sea’ of electrons helps to bond the metallic ions tightly in a packed layer formation. • It requires a large amount of energy to break this formation apart.
Metallic bonding to explain properties • Use what you know about metallic bonding to explain: • Why metals have a high melting point. • Why metals conduct electricity. • Why metals conduct heat. • How metals are malleable.
Properties explained • High melting point is due to the strong attraction between metal ions and free electrons. • Conductivity due to the electrons ability to move quickly and pass on the energy.
metals are very malleable, they can be readily bent, pressed or hammered into shape. The layers of atoms can slide over each other without fracturing the structure The reason for this is the mobility of the electrons.
Alloys • What is an alloy? • An alloy is a mixture of a metal with another element. • This effects the properties of the material by changing the structure. • Make it harder.
Shape memory alloys • What do you think they do? • What could we use them for?
Polymer properties • What are polymers? • What do we use them for? • What are monomers?
Some polymers are made up of more than one type of monomer. • How might this effect their properties? Why? • Another way of changing the properties of a polymer involves changing the conditions of the reaction making the polymer. • Make it hotter, add a catalyst.
The structure of plastics • The lines represent the bonding in the polymer. • Which do you think will be stronger? Why? • Thermosetting plastic • Thermo softening plastics
THERMOSETTING PLASTICS • Once heated and shaped will set. Further heating will not result in a change in shape. • Due to strong cross links between polymer chains. • THERMOSOFTENING PLASTICS • Can be heated and re-shaped. • Once cooled, they can be reheated and reshaped. This can be done as many times as required. • Polymers chains not linked but tangled together.
Give some examples of materials that are made out of the different types of polymers. Thermo softening Thermosetting
Nano-Science Buckmisterfullerene organophosphorus hydrolase (OPH)
One million nanoparticles placed side by side would span 1mm. What is a nanoparticle? GCSE Science Chapter 6
Macro….. Eye 2 metre GCSE Science Chapter 6 Person
Macro….. Eye 20 cm GCSE Science Chapter 6 Science textbook
Macro….. Eye 2 cm GCSE Science Chapter 6 Your finger
Micro….. microscope 300 micrometer GCSE Science Chapter 6 Flea
Micro….. microscope 40 micrometer GCSE Science Chapter 6 Human hair
20 micrometer Micro….. microscope GCSE Science Chapter 6 One blood cell
4 micrometer Micro….. microscope GCSE Science Chapter 6 One Bacterium E-coli
400 nanometre Electron microscope Nano….. GCSE Science Chapter 6 One Flu Virus (Influenza)
1 nanometre Electron microscope Nano….. GCSE Science Chapter 6 Molecule of Carbon 60
What is nanoscience? A nanometer (nm) is 1,000,000,000 (1 billion) times smaller than a meter Nano-science is the study of particles that have the size of 1 to100nm Your finger nails grew a nm while reading this!
Imagine a nanoparticle was the size of a football. A chicken would be the size of earth A flea would be the size of Swansea. A virus would be the same size as a human!
Is nano-science about in our lives today? – YES!! • Lots and lots of things that you have used this morning have used nanoscience – straightners, toothpaste, phones, makeup……. • Silver is very widely used - nano sized silver particles have antibacterial, antiviral and antifungal properties. • Where could these be used?
Some socks have been developed with the silver in to help people who suffer from athletes foot Antifungal Properties
Some fridges have the nanosized silver in the coating inside fridges. This will kill bacteria which can cause food poisioning Antibacterial
Nano-sized zinc oxide absorb and reflect UV light while being transparent to visible light. Why is this useful? Where could be use this?
THE BEACH!!! • We use it in sunglasses – UV light damages our eyes! • UV light can cause skin cancer, so we u use it in sun lotion!
Cosmetics Nanaoparticles in face creams can be absorbed much deeper into the skin.
Nano-Gold Nano-cages of gold can be used to deliver drugs to where they need to go in the body. This could be particularly effective in destroying tumour cells. rat's cerebral cortex before and after gold nanocage administration. Increased absorption was shown
Future Developments Nanowires Are being developed and they could be used to make tiny electrical circuits. They could also be used to help make computers with improved memory and speed
Areas of concern What problems could nanoparticles cause?
Homework • Nanotechnology: Fact, fiction and future Task A nanotechnology company has asked you to design a poster or leaflet to help people understand the benefits of nanotechnology. The poster or leaflet must include: • Introduction – explain what nanotechnology is. • FACTS: Identify what nanotechnology is used for now and its benefits. • FICTION: Identify and explain away some of the exaggerated ideas about nanotechnology. • FUTURE: Identify and explain some of the future uses of nanotechnology. Use the grade ladder to help you reach your target grade.