Metallic Bonding
Metallic Bonding. Metallic Bonding. Observations of the physical properties of metals have led chemists to develop theories to explain these observations. Physical Properties of Metals. For example, electrical conductivity requires the presence of charged particles that are free to move.

Metallic Bonding
E N D
Presentation Transcript
Metallic Bonding • Observations of the physical properties of metals have led chemists to develop theories to explain these observations.
Physical Properties of Metals • For example, electrical conductivity requires the presence of charged particles that are free to move. • Hardness and high melting points imply strong bonding. • Malleability and ductility suggest that there is regularity in the structure. • From this information, chemists have devised a model to explain and represent the structure and bonding in metal elements.
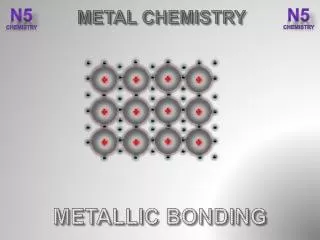
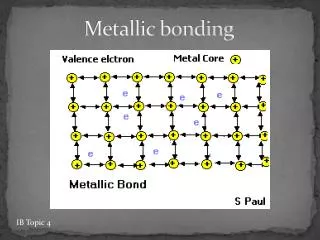
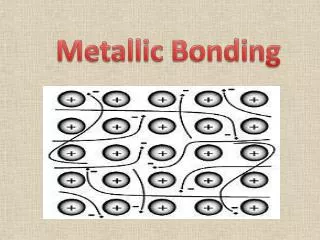
The Nature of the Metallic Bond • In this model, metal ions, formed when atoms lose their valence electrons, are arranged in a three-dimensional lattice. • This array of ions is surrounded by freely moving electrons that form a “sea” of mobile electrons. • These electrons are said to be delocalized, as they are not confined to a particular location but can move throughout the structure.
The Nature of the Metallic Bond • Electrons are attracted to positively charged ions. • This electrostatic attraction holds the lattice together, and prevents the ions pushing each other apart due to the electrostatic repulsion of like charges. • This type of bonding is called metallic bonding.
The Nature of the Metallic Bond • Why do the metal atoms release their valence electrons to form the sea of electrons? • Metal atoms achieve greater stability by releasing their valence electrons. • Without their valence electrons, the metal atoms achieve a noble gas configuration.
The Nature of the Metallic Bond • When non-metals are present, these valence electrons are transferred to the non-metal atoms, giving rise to the ionic bonding discussed last class. • When only metal atoms are present, the “lost” valence electrons simply become delocalized within the metallic lattice.
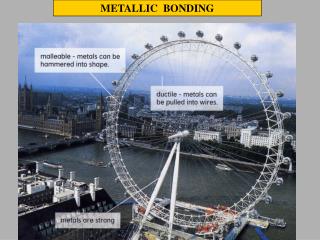
Important Properties of Metals • The uses of metals by humans in the past as well as the present centre on two important properties of metals: • Their ability to conduct electricity • Their malleability
Electrical Conductivity • The electrical conductivity can be explained by the presence of the sea of delocalized electrons that surrounds the lattice of positive metal ions. • In the solid state these electrons can move freely and will respond to the application of a potential difference. • When a metal is connected to a power supply, electrons enter one end of the metal and the same number of electrons exits from the other end of the metal.
Malleability • The malleability of a metal is its ability to be beaten or bent into shape without breaking. • Once again the sea of delocalized electrons is responsible for this property of metals. • When a metal is bent, its lattice of positive ions is displaced and there is a possibility of positive ions coming into contact with other positive ions and repelling each other.
Malleability • The constant movement of the delocalized electrons prevents this from occurring, so the metal bends without breaking.