Metallic Bonding
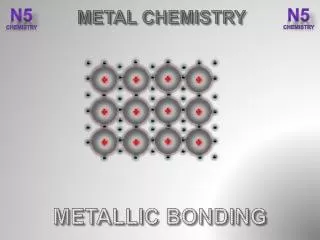
Metallic Bonding. Metallic Bonds. The bonding in metals is explained by the electron sea model , which proposes that the atoms in a metallic solid contribute their valence electrons to form a “sea” of electrons that surrounds metallic cations. . Metallic Bonds.

Metallic Bonding
E N D
Presentation Transcript
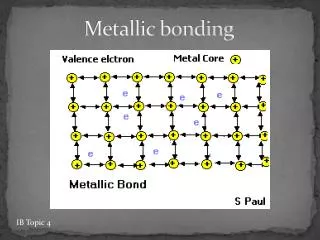
Metallic Bonds The bonding in metals is explained by the electron sea model, which proposes that the atoms in a metallic solid contribute their valence electrons to form a “sea” of electrons that surrounds metallic cations.
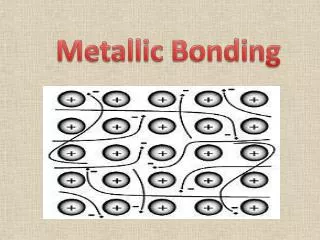
Metallic Bonds • These delocalized electrons are not held by any specific atom and can move easily throughout the solid. • Metal atoms release their valence electrons into a sea of electrons shared by all of the metal atoms. The bond that results from this shared pool of valence electrons is called a metallicbond.
Metallic Bonds Metals hold onto their valence electrons very weakly. Think of them as positive ions floating in a sea of electrons.
Metallic Bond Properties + + + + + + + + + + + + Electrons are free to move through the solid. Metals conduct electricity.
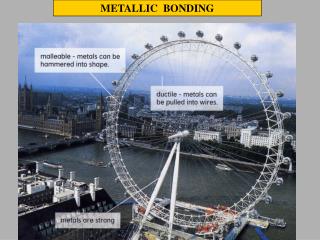
Metallic Bond Properties Metals generally have extremely high melting points because it is difficult to pull metal atoms completely away from the group of cations and attracting electrons.
Metallic Bond Properties Metals are malleable (able to be hammered into sheets).
Metallic Bond Properties Metals are also ductile (able to be drawn into wire) because of the mobility of the particles.
Malleable + + + + + + + + + + + +
Malleable + + + + + + + + + + + + Electrons allow atoms to slide by.
Alloys A mixture of elements that has metallic properties is called an alloy.