Metallic Bonding
CHEMISTRY CHAPTER 6 SECTIONS 4-5 SECTION 4. METALLIC BONDING. Metallic Bonding Chemical bonding is different in metals than it is in ionic or molecular compounds. Characteristic properties of metals: electrical conductivity thermal conductivity malleability ductility shiny appearance.

Metallic Bonding
E N D
Presentation Transcript
CHEMISTRY CHAPTER 6 SECTIONS 4-5 SECTION 4. METALLIC BONDING Metallic Bonding • Chemical bonding is different in metals than it is in ionic or molecular compounds. • Characteristic properties of metals: • electrical conductivity • thermal conductivity • malleability • ductility • shiny appearance
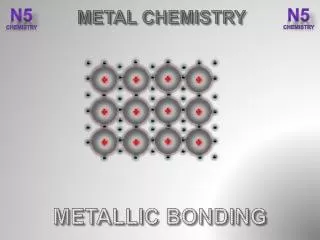
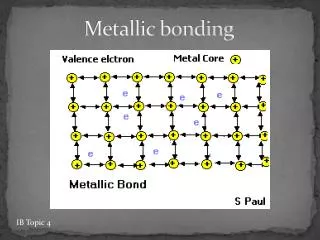
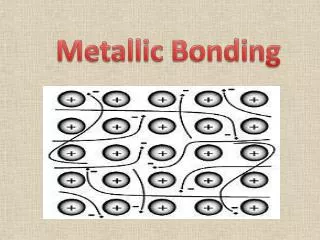
The Metallic-Bond Model • In a metal, the vacant orbitals in the atoms’ outer energy levels overlap. • This overlapping of orbitals allows the outer electrons of the atoms to roam freely throughout the entire metal.
The electrons are delocalized,which means that they do not belong to any one atom but move freely about the metal’s network of empty atomic orbitals. • These mobile electrons form a sea of electronsaround the metal atoms, which are packed together in a crystal lattice.
The chemical bonding that results from the attraction between metal atoms and the surrounding sea of electrons is calledmetallic bonding.
Metallic Properties 1. Since electrons move easily, there is high thermal and electrical conductivity. 2. There are many orbitals with small energy differences. Electrons can absorb light, and then re-radiate it. This causes luster (the shiny metallic appearance).
3. Bonding is the same in all directions, so it is easy to slide layers of atoms past each other. This leads to the malleability and ductility of metals. (How do ionic compounds compare?)
Malleabilityis the ability of a substance to be hammered or beaten into thin sheets. • Ductilityis the ability of a substance to be drawn, pulled, or extruded through a small opening to produce a wire.
Properties of Substances with Metallic, Ionic, and Covalent Bonds
Properties of Metals: Electrical and Thermal Conductivity – 75131Surface Appearance – 75132Malleability and Ductility - 75133
SECTION 5. MOLECULAR GEOMETRY The properties of molecules depend not only on the bonding of atoms but also on molecular geometry:the three-dimensional arrangement of a molecule’s atoms.
The polarity of each bond, along with the geometry of the molecule, determinesmolecular polarity,or the uneven distribution of molecular charge. • Molecular polarity strongly influences the forces that act between molecules in liquids and solids.
VSEPR Theory • Molecular geometry can be predicted using VSEPR theory(= valence-shell, electron pair repulsion). • The theory states that repulsion between sets of valence-level electrons around an atom causes them to be oriented as far apart as possible.
Beginning with molecules where the central atom has no unshared pairs of electrons: the bonds will be as far apart as possible. Example 1: BeF2 • Lewis structure: • The bonds to fluorine will be 180° apart from each other, so the molecule is linear. wikipedia
Example 2: BCl3 • Lewis structure: • The bonds are 120° apart. • The shape of the molecule is trigonal-planar. wikipedia
Example 3: CH4 • Lewis structure: • The bonds are arranged in a tetrahedral shape. cnx.org citycollegiate.com
Now consider molecules with unshared electron pairs (lone pairs): the lone pairs occupy space and also repel other pairs of electrons (such as those in bonds). After you determine the orientations of the bonds and lone pairs in space, keep in mind that the shape of the molecule refers only to the positions of the atoms.
Example 1: ammonia (NH3) The central nitrogen atom has an unshared electron pair: • VSEPR theory postulates that the lone pair occupies space around the nitrogen atom just as the bonding pairs do.
The four pairs of electrons have approximately a tetrahedral arrangement. The shape of the molecule is a pyramid with a triangular base.
Unshared pairs of electrons repel more than pairs in bonds, so the angles are not all the same. chemwiki.ucdavis.edu
Example 2: H2O. The oxygen atom has two unshared electron pairs: H – O – H The bonds and lone pairs take approximately a tetrahedral arrangement. The shape of the molecule is bent.
Other shapes Some other shapes can be found when the central atom has more than an octet: • Trigonal bipyramid. Ex. – PCl5 chem.ufl.edu elmhurst.edu
2. Octahedral. Ex. – SF6 uwplatt.edu chemwiki.ucdavis.edu
3. Square planar. Ex. – XeF4 uwplatt.edu schools-wikipedia.org
Multiple bonds are treated like single bonds in VSEPR. Examples: CO2 and HCN – there are no more electron pairs in the central atom so the molecules are linear. O = C = O H – C N
Molecular Polarity A polar covalent bond has a slight negative (δ-) charge on one end and a slight positive (δ+) charge on the other. (δ = lower case Greek letter delta) chem.fsu.edu
Molecular Polarity If the charges from polar bonds are not symmetrically arranged in space, the molecule will be polar. δ- δ+ chem.fsu.edu
If partial positive and negative charges are arranged symmetrically in space, the molecule is nonpolar. angelfire.com cdli.ca
Polar molecules are more strongly attracted to each other than nonpolar, so they have higher melting and boiling points. • Ex. – H2O boils at 100 °C; CH4 boils at -164 °C Prentice-Hall Physical Science