Understanding Metallic Bonding: Properties and Alloy Examples
Metallic bonding results from the attraction between metal atoms and a 'sea of electrons', which allows electrons to move freely within the structure while the metal atoms remain fixed in rigid positions. This electron mobility imparts unique properties to metals, including luster, malleability, ductility, and excellent conductivity of heat and electricity. Common alloys like Alnico, brass, bronze, and stainless steel illustrate the combination of different metals, enhancing desirable characteristics. Notably, while most metals are solid at room temperature, mercury is an exception.

Understanding Metallic Bonding: Properties and Alloy Examples
E N D
Presentation Transcript
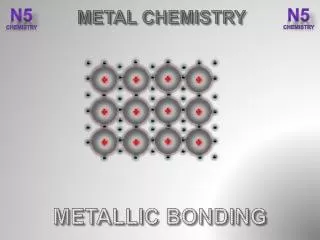
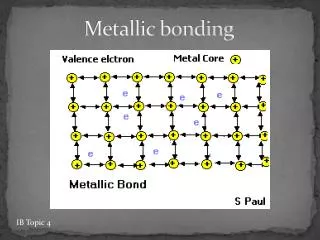
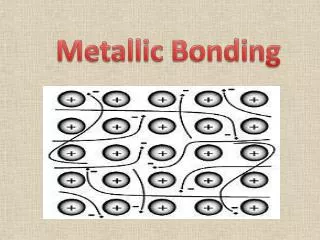
METALLIC BONDING Results from the attraction between metal atoms and the surrounding “SEA OF ELECTRONS”
In Metallic Bonding the atoms are in a FIXED, RIGID position while the electrons are free to move throughout the crystal. The “d” and “p” orbitals overlap each other since not all the “d” are completely filled and the some of the “p” are also vacant. These overlapping electrons are MOBILE, free to move.
This mobility of the “sea of electrons” accounts for the metallic properties.
Properties of Metals • Luster, shiny • Malleable • Ductile • Crystalline • Solids at room temperature EXCEPT for Mercury- Hg • Good conductors of heat and electricity • Melting points vary widely
Examples of Alloys See Table on Page 231 Alnico 50%Fe, 20% Al, 20% Ni, 10% Co used in magnets Brass 67-90% Cu, 10-33% Zn Bronze Cu, Zn, Sn Cast Iron Fe and C Dental amalgam Hg, Ag. Sn Gold, 10 carat 42% Au, 12-20 % Ag, 38-46% Cu Pewter Sn, Sb, Pb Stainless steel Fe, Cr, Ni Sterling Silver Ag, Cu
So you could say that an alloy is a SOLUTION OF METALS