METALLIC BONDING
170 likes | 421 Views
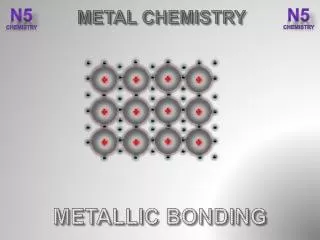
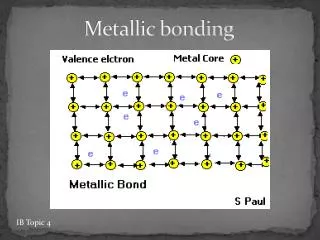
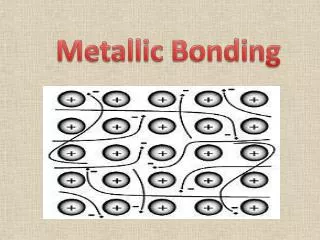
METALLIC BONDING. METALLIC BONDING. Metals conduct heat and electricity. Metals have these properties because of the way their atoms are joined together. METALLIC BONDING. metal ion. mobile electrons (de-localised). Metals consist of ‘Giant structures’.

METALLIC BONDING
E N D
Presentation Transcript
METALLIC BONDING Metals conduct heat and electricity Metals have these properties because of the way their atoms are joined together.
METALLIC BONDING metal ion mobile electrons (de-localised) Metals consist of ‘Giant structures’. The electrons from outer energy levels are free to move through the whole structure . These electrons * hold the atoms together ( + ions attract - electrons) * allow the atoms to slide over each other (metals bend) * allow the metal to conduct heat and electricity
METALLIC BONDING The atoms in a metal are closely packed so …………………………………….. The atoms are in a regular pattern so …………………………………….. Metallic bonding is strong so ……………………………………... PHRASE LIST: metals have high melting points metals are dense they have a crystal structure
METALLIC BONDING The atoms in a metal are closely packed so metals are dense. The atoms are in a regular pattern so they have a crystal structure. Metallic bonding is strong so metals have high melting points PHRASE LIST: metals have high melting points metals are dense they have a crystal structure
METALLIC BONDING Galvanised dustbin The crystals on a metal’s surface are called grains. Crystals join together at grain boundaries
If cooled slowly larger crystals grow
giant electrons outer positive electrostatic free Cool liquid metal slowly to increase crystal size Smaller grains give a harder but more brittle metal The de-localised electrons are like glue because: they attract the positive metal ions (hold them together) They are not like glue because: they are able to move through the whole lattice
METALLIC BONDING The crystals on a metal’s surface are called grains. Crystals join together at grain boundaries
METALLIC BONDING Small grains tend to stop rows of atoms sliding over each other. This makes the metal harder but more brittle. Zinc ion Brass Copper ion
METALLIC BONDING Zinc ion Brass Copper ion