LECTURE 5: PHASE EQUILIBRIA
LECTURE 5: PHASE EQUILIBRIA. PHASE EQUILIBRIA. Phase equilibrium describes the way phases (such as solid, liquid and/or gas) co-exist at some temperatures and pressure, but interchange at others. Energetic introduction to phase equilibria. Why does an ice cube melt in the mouth?.

LECTURE 5: PHASE EQUILIBRIA
E N D
Presentation Transcript
PHASE EQUILIBRIA Phase equilibrium describes the way phases (such as solid, liquid and/or gas) co-exist at some temperatures and pressure, but interchange at others.
Energetic introduction to phase equilibria Why does an ice cube melt in the mouth?
Further thermodynamic background: terminology A phaseis a component within a system, existing in a precisely defined physical state, e.g. gas, liquid, or a solid that has a single crystallographic form. Concerning transitions between the two phases ‘1’ and ‘2’, Hess’s Law states that H(1→2) = −1 × H(2→1).
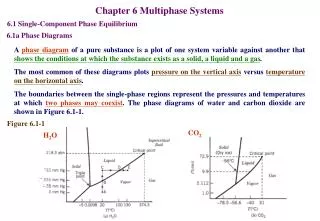
PHASE TERMINOLOGY A phase diagram is a graph showing values of applied pressure and temperature at which equilibrium exists. A phase boundary is a line on a phase diagram representing values of applied pressure and temperature at which equilibrium exists. The triple point on a phase diagram represents the value of pressure and temperature at which three phases coexist at equilibrium.
SPONTANEITY OF PHASE CHANGES Why does water placed in the freezer become ice?
SOLID PHASE TRANSITIONS the tin allotropes have very different densities ρ, so ρ(tin, grey) = 5.8 gcm−3 but ρ(tin, white) = 7.3 gcm−3.
KINETICS OF PHASE CHANGES Phase transitions involving liquids and gases are generally fast!
Pressure and temperature changes with a single-component system: How is the ‘Smoke’ in horror films made? The effect of temperature on phase change: sublimation
The Effect of Pressure on Phase Change: How does freeze drying work? Freeze-drying is a layman’s description, and acknowledges that external conditions may alter the conditions of a phase change, i.e. the drying process (removal of water) occurs at a temperature lower than 100 ◦C.
THERMODYNAMICS OF PHASE CHANGES HOW DOES A ROTARY EVAPORATOR WORKS?
CRITICAL AND SUPERCRITICAL FLUIDS How is coffee decaffeinated?
COFFEE DECAFFEINATED The intensive properties of the liquid and gas (density, heat capacity, etc.) become equal at the critical point, which is the highest temperature and pressure at which both the liquid and gaseous phases of a given compound can coexist.
QUANTITATIVE ASPECTS OF P-T CHANGE Why is ice so slippery? The effect of p and T on the position of solid liquid equilibrium
P-T Quantitative Relationship Clapeyron Equation:
SAMPLE PROBLEM: Consider a car weighing 1000 kg (about 2200 lbs) parked on a sheet of ice at 273.15 K. Take the area under wheels in contact with the ice as 100 cm2 i.e. 10−2 m2. What is the new melting temperature of the ice – call it T(final)? Take H O(melt) = 6.0 kJmol−1 and water Vm(melt) = −1.6 × 10−6 m3 mol−1.
SAMPLE PROBLEM Paraffin wax has a normal melting temperature T(melt) of 320 K. The temperature of equilibrium is raised by 1.2 K if the pressure is increased fivefold. Calculate Vm for the wax as it melts. Take H(melt) = 8.064 kJ mol−1.
PHASE DIAGRAMS • knowledge of the thermodynamics of simple mixtures to discuss the physical changes of mixtures when they are heated or cooled and when their compositions are changed. • phase diagrams can be used to judge whether two substances are mutually miscible.
PHASE DIAGRAMS • can equilibrium exist over a range of conditions or whether a system must be bought to a definite pressure, temperature and composition before equilibrium is established. • phase diagrams are industrially and commercially important.
PHASE DIAGRAMS • semiconductor, ceramics, steel and alloy industries rely heavily on phase diagrams to ensure uniformity of a product. • phase diagrams are also the basis for separation procedures in the petroleum industry and the formulation of foods and cosmetic preparations.
DEFINITIONS • A phase is a state of matter that is uniform throughout, not only in composition but also in physical state. • A pure gas • A gaseous mixture • Two totally miscible liquids • A crystal
DEFINITIONS • A solution of sodium chloride • Ice • A slurry of ice and water
DEFINITIONS • An alloy of two metals?
DEFINITIONS • an alloy of two metals is a two phase system if the metals are immiscible, but a single phase system if they are miscible. • dispersion can be uniform on a macroscopic level, but not on a microscopic scale. • dispersions are important in many advanced materials.
DEFINITIONS • heat treatment cycles are used to achieve the precipitation of a fine dispersion of particles of one phase within a matrix formed by a saturated solid solution phase. • the ability to control this microstructure resulting from phase equilibria makes it possible to tailor the mechanical properties of the materials.
DEFINITIONS • A constituent of a system is a chemical species (an ion or a molecule) that is present. • A mixture of water and ethanol has two constituents. • A solution of sodium chloride has three constituents: Na+, Cl-, H2O.
DEFINITIONS • acomponent is a chemically independent constituent of a system. • the number of components in a system is the minimum number of independent species necessary to define the composition of all the phases present in the system.
DEFINITIONS • When no reaction takes place and there are no other constraints, the number of components is the equal to the number of constituents. • Pure water is a one component system • A mixture of ethanol and water is two component system.
DEFINITIONS • an aqueous solution of sodium chloride is a two component system, because by charge balance, the number of Na+ ions must be the same as the number of Cl- ions. • a system that consists of hydrogen, oxygen and water at room temperature has three components.
DEFINITIONS • when a reaction can occur under the conditions prevailing in the system, we need to decide the minimum number of species that, after allowing for reactions in which one species is synthesized from others, can be used to specify the composition of all the phases.
DEFINITIONS • CaCO3(s) CaO(s) + CO2(g) • 3 phases • 3 constituents • To specify the composition of the gas phase, we need the species CO2, and to specify the composition of the solid phase on the right, we need the species CaO.
DEFINITIONS • CaCO3(s) CaO(s) + CO2(g) • We do not need an additional species to specify the composition of the phase on the right, because its identity (CaCO3) can be expressed in terms of the other two constituents by making use of the stoichiometry of the reaction. • 2 component system.
SAMPLE PROBLEM How many components are present in a system in which ammonium chloride undergoes thermal decomposition? The reaction is: NH4Cl(s) NH3(g) + HCl(g)
DEFINITIONS • NH4Cl(s) NH3(g) + HCl(g) • 2 phases • 3 constituents • 1 component
SAMPLE PROBLEM • Give the number of components in the following systems: (a) water, allowing for its autoprotolysis, (b) aqueous acetic acid, (c) magnesium carbonate in equilibrium with its decomposition products.
DEFINITIONS • The number of phases, P. • The number of components, C. • The variance of the system, F is the number of intensive variables (e.g. p and T) that can be changed independently without disturbing the number of phases in equilibrium.
PHASE RULE • F = C – P + 2 • This is not an empirical rule based upon observations, it can be derived from chemical thermodynamics • For a one component system F = 3 – P • When only one phase is present, F = 2 and both p and T can be varied without changing the number of phases.
PHASE RULE • When two phases are present, F = 1 which implies that pressure is not freely variable if the pressure is set. This is why at a given temperature a liquid has a characteristic vapor pressure. • When three phases are present, F = 0. This special case occurs only at a definite temperature and pressure that is characteristic of the substance.
EXPERIMENTAL PROCEDURE • Thermal analysis – a sample is allowed to cool and it temperature is monitored. When a phase transition occurs, cooling may stop until the phase transition is complete and is easily observed on a thermogram.