VLE Calculations
VLE Calculations. Purpose of lecture : To demonstrate how Raoult’s law is used to predict VLE behaviour of ideal mixtures Highlights Phase rules gives the number of variables neede to determine the intensive state of a system at equilibrium

VLE Calculations
E N D
Presentation Transcript
VLE Calculations • Purpose of lecture: • To demonstrate how Raoult’s law is used to predict VLE behaviour of ideal mixtures • Highlights • Phase rules gives the number of variables neede to determine the intensive state of a system at equilibrium • Saturation pressures can be calculated by means of the Antoine Eqn. • Raoult’s law can be used for constructing Pxy, Txy diagrams and performing dew point and bubble point calculations • Reading assignment: Section 10.4, pp. 347-357 (7th edition), or • Section 10.4, pp. 338-348 (6th edition) Lecture 2
Gibbs Phase Rule for Intensive Variables SVNA-10.2 • For a system of phases and N species, the degree of freedom is: • F = 2 - + N • F = # of variables that must be specified to fix the intensive state of the system at equilibrium • Phase Rule Variables: • The system is characterized by T, P and (N-1) mole fractions for each phase • 2 + (N-1) variables must be specified • Phase Rule Equations: • At equilibrium i = i = i for all N species • (-1)N independent equations can be written in terms of T, P and compositions • Degrees of freedom: F = 2 + (N-1) - (-1)N • = 2- +N Lecture 2
Phase Rule in VLE: Single Component Systems • For a two phase (p=2) system of a single component (N=1): • F = 2- + N • F = 2- 2 + 1 = 1 • Therefore, for the single component system, specifying either T or P fixes all intensive variables. List some of them. Lecture 2
Correlation of Vapour Pressure Data • Pisat, or the vapour pressure of component i, is commonly represented by Antoine Equation (Appendix B, Table B.2, SVNA 7th ed.): • For acetonitrile (Component 1): • For nitromethane (Component 2): • These functions are the only component properties needed to characterize ideal VLE behaviour Lecture 2
Phase Rule in VLE: Ideal Binary Mixtures • (General Case) • For a two phase (=2), binary system (N=2): • F = 2 - + N = 2 • Therefore, for the binary case, two intensive variables must be specified to fix the state of the system. How does this work? Lecture 2
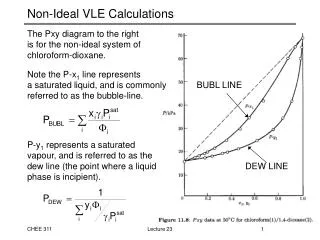
Phase Rule in VLE: Binary Systems (Pxy diagrams) • Example: Acetonitrile (1) / Nitromethane (2) system Which component is more volatile? What phases are present in each region? Lecture 2
Phase Rule in VLE: Binary Systems (Txy diagrams) • Alternatively, we can specify a system pressure and examine the VLE behaviour as a function of temperature and composition. What phases are present in each region? What would this all look like in 3D? Lecture 2
VLE Calculations using Raoult’s Law • Raoult’s Law for ideal phase behaviour relates the composition of liquid and vapour phases at equilibrium through the component vapour pressure, Pisat. • Given the appropriate information, we can apply Raoult’s law to the solution of 5 types of problems: • Dew Point: Pressure or Temperature • Bubble Point: Pressure or Temperature • P,T Flash: calculation of equilibrium composition (P, T, zi given) What is zi? Lecture 2
Dew and Bubble Point Calculations • Dew Point Pressure: • Given a vapour composition at a specified temperature, find the composition of the liquid in equilibrium • Given T, y1, y2,... yn find P, x1, x2, ... xn • Dew Point Temperature: • Given a vapour composition at a specified pressure, find the composition of the liquid in equilibrium • Given P, y1, y2,... yn find T, x1, x2, ... xn • Bubble Point Pressure: • Given a liquid composition at a specified temperature, find the composition of the vapour in equilibrium • Given T, x1, x2, ... xn find P, y1, y2,... yn • Bubble Point Temperature: • Given a vapour composition at a specified pressure, find the composition of the liquid in equilibrium • Given P, x1, x2, ... xn find T, y1, y2,... yn • Why these names? Lecture 2
VLE Calculations - Introduction • For now, we will do calculations only for binary and ideal mixtures • Multicomponent nonideal situations later • The calculations use two key equations: • 1) Raoult’s law for ideal phase behaviour: • 2) Antoine Equation for vapour pressures of pure components (1) (2) Lecture 2
BUBL P Calculation (T, x1 known) • What do we want to find out? • How do we do it? • What about BUBL T, DEW P, DEW T? Lecture 2
Example • Assuming Raoult’s Law to be valid, prepare • a Pxy diagram for T=90oC, and • a Txy diagram for P=90 kPa • for a mixture of 1-chlorobutane (1) /chlorobenzene (2) • Antoine Coefficients: Let’s list the steps required. How could we do it using a spreadsheet? Lecture 2
Example – Generation of Txy Diagram Lecture 2
Example – (b) Construction of a Txy Plot Lecture 2
VLE Calculations - Summary • Why? • How? • Who cares? • Which type is the most difficult? Lecture 2