VLE Calculations
VLE Calculations. Purpose of this lecture : To demonstrate how Raoult’s law can be used in the prediction of the VLE behaviour of ideal mixtures Highlights Phase rules gives the number of variables we need in order to determine the intensive state of a system at equilibrium

VLE Calculations
E N D
Presentation Transcript
VLE Calculations • Purpose of this lecture: • To demonstrate how Raoult’s law can be used in the prediction of the VLE behaviour of ideal mixtures • Highlights • Phase rules gives the number of variables we need in order to determine the intensive state of a system at equilibrium • Saturation pressures can be calculated by means of the Antoine Eqn. • Raoult’s law can be used for constructing Pxy, Txy diagrams and performing dew point and bubble point calculations • Reading assignment: Section 10.4, pp. 347-357 (7th edition), or • Section 10.4, pp. 338-348 (6th edition) Lecture 2
Phase Rule for Intensive Variables SVNA-10.2 • For a system of phases and N species, the degree of freedom is: • F = 2 - + N • # variables that must be specified to fix the intensive state of the system at equilibrium • Phase Rule Variables: • The system is characterized by T, P and (N-1) mole fractions for each phase • Requires knowledge of 2 + (N-1) variables • Phase Rule Equations: • At equilibrium i = i = i for all N species • These relations provide (-1)N equations • The difference is F = 2 + (N-1) - (-1)N • = 2- +N Lecture 2
Phase Rule in VLE: Single Component Systems • For a two phase (p=2) system of a single component (N=1): • F = 2- + N • F = 2- 2 + 1 = 1 • Therefore, for the single component system, specifying either T or P fixes all intensive variables. Lecture 2
Correlation of Vapour Pressure Data • Pisat, or the vapour pressure of component i, is commonly represented by Antoine Equation (Appendix B, Table B.2, SVNA 7th ed.): • For acetonitrile (Component 1): • For nitromethane (Component 2): • These functions are the only component properties needed to characterize ideal VLE behaviour Lecture 2
Phase Rule in VLE: Ideal Binary Mixtures • (General Case) • For a two phase (=2), binary system (N=2): • F = 2- 2 + 2 = 2 • Therefore, for the binary case, two intensive variables must be specified to fix the state of the system. Lecture 2
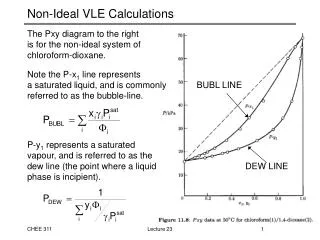
Phase Rule in VLE: Binary Systems (Pxy diagrams) • Example: Acetonitrile (1) / Nitromethane (2) system Lecture 2
Phase Rule in VLE: Binary Systems (Txy diagrams) • Alternatively, we can specify a system pressure and examine the VLE behaviour as a function of temperature and composition. Lecture 2
VLE Calculations using Raoult’s Law • Raoult’s Law for ideal phase behaviour relates the composition of liquid and vapour phases at equilibrium through the component vapour pressure, Pisat. • Given the appropriate information, we can apply Raoult’s law to the solution of 5 types of problems: • Dew Point: Pressure or Temperature • Bubble Point: Pressure or Temperature • P,T Flash: calculation of equilibrium composition (P, T, zi given) Lecture 2
Dew and Bubble Point Calculations • Dew Point Pressure: • Given a vapour composition at a specified temperature, find the composition of the liquid in equilibrium • Given T, y1, y2,... yn find P, x1, x2, ... xn • Dew Point Temperature: • Given a vapour composition at a specified pressure, find the composition of the liquid in equilibrium • Given P, y1, y2,... yn find T, x1, x2, ... xn • Bubble Point Pressure: • Given a liquid composition at a specified temperature, find the composition of the vapour in equilibrium • Given T, x1, x2, ... xn find P, y1, y2,... yn • Bubble Point Temperature: • Given a vapour composition at a specified pressure, find the composition of the liquid in equilibrium • Given P, x1, x2, ... xn find T, y1, y2,... yn Lecture 2
VLE Calculations - Introduction • For now, we are going to employ these calculations only for • identifying the state and composition of binary and ideal mixtures • As we are going to see later in the course, the aforementioned • VLE calculations are also applicable to non-ideal or/and • multi-component mixtures • The calculations revolve around the use of 2 key equations: • 1) Raoult’s law for ideal phase behaviour: • 2) Antoine’s Equation (1) (2) Lecture 2
BUBL P Calculation (T, x1 known) • - Calculate and from Antoine’s Equation • For the vapour-phase composition (bubble) we can write: • y1+y2=1 (3) • Substitute y1 and y2 in Eqn (3) by using Raoult’s law: • (4) • - Re-arrange and solve Eqn. (4) for P • Now you can obtain y1 from Eqn (1) • Finally, y2 = 1-y1 Lecture 2
DEW P Calculation (T, y1 known) • - Calculate and from Antoine’s Equation • For the liquid-phase composition (dew) we can write: • x1+x2=1 (5) • Substitute x1 and x2 in Eqn (5) by using Raoult’s law: • (6) • - Re-arrange and solve Eqn. (6) for P • Now you can obtain x1 from Eqn (1) • Finally, x2 = 1-x1 Lecture 2
BUBL T Calculation (P, x1 known) • Since T is an unknown, the saturation pressures for the • mixture components cannot be calculated directly. Therefore, • calculation of T, y1 requires an iterative approach, as follows: • Re-arrange Antoine’s equation so that the saturation temperatures • of the components at pressure P can be calculated: • (7) • Select a temperature T’ so that • Calculate • Solve Eqn. (4) for pressure P’ • If , then P’=P; If not, try another T’-value • Calculate y1 from Raoult’s law Lecture 2
DEW T Calculation (P, y1 known) • Same as before, calculation of T, x1 requires an iterative approach: • Re-arrange Antoine’s equation so that the saturation temperatures • of the components at pressure P can be calculated from Eqn. (7): • - Select a temperature T’ so that • Calculate from Antoine’s Eqn. • Solve Eqn. (6) for pressure P’ • If , then P’=P; If not, try another T’-value • Calculate x1 from Raoult’s law Lecture 2
P, T Flash Calculation • - Calculate and from Antoine’s Equation • Use Raoult’s law in the following form: • (8) • - Re-arrange and solve Eqn. (8) for x1 • Now you can obtain y1 from Eqn (1), i.e., Lecture 2
Example • Assuming Raoult’s Law to be valid, prepare • a Pxy diagram for T=90oC, and • a Txy diagram for P=90 kPa • for a mixture of 1-chlorobutane (1) /chlorobenzene (2) • Antoine Coefficients: Lecture 2
Construction of Pxy diagrams • The construction of Pxy diagram requires multiple P, T Flash • calculations, where T is held constant and P is varied from P2sat to P1sat. • The results can be tabulated as shown below: This type of calculations can also be performed by keeping T constant and varying x1 or y1 from 0.0 to 1.0 Lecture 2
Example* – (a) Generation of Pxy Data Lecture 2
160.00 140.00 120.00 liquid 100.00 x1 P (kPa) 80.00 y1 VLE 60.00 40.00 vapor 20.00 0.00 0.00 0.20 0.40 0.60 0.80 1.00 Example – (a) Construction of a Pxy Plot Lecture 2
Construction of Txy diagrams • The construction of Txy, diagram requires multiple P, T, Flash • calculations, each one of which provides a set of equilibrium y1, x1 • values for a given value of temperature (at fixed P) • The results can be tabulated as shown below: This type of calculations can also be performed by keeping P constant and varying x1 or y1 from 0.0 to 1.0 Lecture 2
Example – (b) Generation of Txy Data Lecture 2
Example – (b) Construction of a Txy Plot Lecture 2
VLE Calculations - Summary • Why? To completely identify the thermodynamic state • of a mixture at equilibrium (single phase, 2 phases..?) • How? Through the calculation of its P, T, and composition • - The type of calculation that we need to perform is subject • to the variables we are looking to evaluate • - These calculations are classified as follows: Lecture 2