EXPLORING PROTEIN STRUCTURE
EXPLORING PROTEIN STRUCTURE. A teaching tool for introducing students to protein structure. A protein hormone which helps to regulate your blood sugar levels. Proteins…. If there is a job to be done in the molecular world of our cells, usually that job is done by a protein.

EXPLORING PROTEIN STRUCTURE
E N D
Presentation Transcript
EXPLORING PROTEIN STRUCTURE A teaching tool for introducing students to protein structure.
A protein hormone which helps to regulate your blood sugar levels Proteins…. If there is a job to be done in the molecular world of our cells, usually that job is done by a protein. Examples of proteins include hormones acting as messengers; enzymes speeding up reactions; cell receptors acting as ‘antennae’; antibodies fighting foreign invaders; membrane channels allowing specific molecules to enter or leave a cell; they make up the muscles for moving; let you grow hair, ligaments and fingernails; and let you see (the lens of your eye is pure crystalised protein). CATALASE An enzyme which removes Hydrogen peroxide from your body so it does not become toxic Source: http://courses.washington.edu/conj/protein/insulin2.gif http://www.biochem.ucl.ac.uk/bsm/pdbsum/1gwf/main.html
Proteins can be fibrous or globular Let’s explore the diversity of protein structure and function by investigating some examples
Fibrous proteins have a structural role • Collagen is the most abundant protein in vertebrates. Collagen fibers are a major portion of tendons, bone and skin. Alpha helices of collagen make up a triple helix structure giving it tough and flexible properties. • Fibroin fibers make the silk spun by spiders and silk worms stronger weight for weight than steel! The soft and flexible properties come from the beta structure. • Keratin is a tough insoluble protein that makes up the quills of echidna, your hair and nails and the rattle of a rattle snake. The structure comes from alpha helices that are cross-linked by disulfide bonds. Source:http://www.prideofindia.net/images/nails.jpghttp://opbs.okstate.edu/~petracek/2002%20protein%20structure%20function/CH06/Fig%2006-12.GIF http://my.webmd.com/hw/health_guide_atoz/zm2662.asp?printing=true
The globular proteins The globular proteins have a number of biologically important roles. They include: 1. Cell motility – proteins link together to form filaments which make movement possible. 2. Organic catalysts in biochemical reactions – enzymes 3. Regulatory proteins – hormones, transcription factors 4. Membrane proteins – MHC markers, protein channels, gap junctions 5. Defense against pathogens – poisons/toxins, antibodies, complement 6. Transport and storage – hemoglobin and myosin
Proteins for cell motility Above: Myosin (red) and actin filaments (green) in coordinated muscle contraction. Right: Actin bound to the mysoin binding site (groove in red part of myosin protein). Add energy (ATP) and myosin moves, moving actin with it. Source: http://www.ebsa.org/npbsn41/maf_home.html http://sun0.mpimf-
Proteins in the Cell Cytoskeleton Eukaryote cells have a cytoskeleton made up of straight hollow cylinders called microtubules (bottom left). They help cells maintain their shape, they act like conveyer belts moving organelles around in the cytoplasm, and they participate in forming spindle fibres in cell division. Microtubules are composed of filaments of the protein, tubulin (top left) . These filaments are compressed like springs allowing microtubules to ‘stretch and contract’. 13 of these filaments attach side to side, a little like the slats in a barrel, to form a microtubule. This barrel shaped structure gives strength to the microtubule. Tubulin forms helical filaments Source:heidelberg.mpg.de/shared/docs/staff/user/0001/24.php3?department=01&LANG=en http://www.fz-juelich.de/ibi/ibi-1/Cellular_signaling/ http://cpmcnet.columbia.edu/dept/gsas/anatomy/Faculty/Gundersen/main.html
No catalyst = Input of 71kJ energy required Energy Activation Energy With catalase = Input of 8 kJ energy required Progress of reaction Proteins speed up reactions - Enzymes 2 + 2 Catalase speeds up the breakdown of hydrogen peroxide, (H2O2) a toxic by product of metabolic reactions, to the harmless substances, water and oxygen. The reaction is extremely rapid as the enzyme lowers the energy needed to kick-start the reaction (activation energy) Substrate Product
Proteins can regulate metabolism – hormones When your body detects an increase in the sugar content of blood after a meal, the hormone insulin is released from cells in the pancreas. Insulin binds to cell membranes and this triggers the cells to absorb glucose for use or for storage as glycogen in the liver. Proteins span membranes –protein channels The CFTR membrane protein is an ion channel that regulates the flow of chloride ions. Not enough of this protein gets inserted into the membranes of people suffering Cystic fibrosis. This causes secretions to become thick as they are not hydrated. The lungs and secretory ducts become blocked as a consequence. Source: http://www.biology.arizona.edu/biochemistry/tutorials/chemistry/page2.html http://www.cbp.pitt.edu/bradbury/projects.htm
Proteins Defend us against pathogens –antibodies Left: Antibodies like IgG found in humans, recognise and bind to groups of molecules or epitopes found on foreign invaders. Right: The binding site of an antigen protein (left) interacting with the epitope of a foreign antigen (green) Source: http://www.biology.arizona.edu/immunology/tutorials/antibody/FR.html http://tutor.lscf.ucsb.edu/instdev/sears/immunology/info/sears-ab.htm http://www.spilya.com/research/ http://www.umass.edu/microbio/chime/
Making Proteins How are such a diverse range of proteins possible? The code for making a protein is found in your genes (on your DNA). This genetic code is copied onto a messenger RNA molecule. The mRNA code is read in multiples of 3 (a codon) by ribosomes which join amino acids together to form a polypeptide. This is known as gene expression. Source: http://genetics.nbii.gov/Basic1.html
M S F T L K E G G E S G G G M E E E L L L T T T F F F S K M G K F T L S E K S T G A C T A G E G M S K S S G G M G G M K E E K K T M M T T G F G F F G G L E L L E E E E AUGAGUAAAGGAGAAGAACUUUUCACUGGAUA E E S G K G M E E L T F The protein folds to form its working shape Gene Expression Gene DNA Cell machinery copies the code making an mRNA molecule. This moves into the cytoplasm. Ribosomes read the code and accurately join Amino acids together to make a protein CELL The order of bases in DNA is a code for making proteins. The code is read in groups of three NUCLEUS Chromosome S K G E M F E G L T
The building blocks The amino acids for making new proteins come from the proteins that you eat and digest. Every time you eat a burger (vege or beef), you break the proteins down into single amino acids ready for use in building new proteins. And yes, proteins have the job of digesting proteins, they are known as proteases. There are only 20 different amino acids but they can be joined together in many different combinations to form the diverse range of proteins that exist on this planet
H S H H H H C C H CH3 H H C H H3C C C C Amino Acids An amino acid is a relatively small molecule with characteristic groups of atoms that determine its chemical behaviour. The structural formula of an amino acid is shown at the end of the animation below. The R group is the only part that differs between the 20 amino acids. Phenylalanine Cysteine Glycine Alanine Valine H R O H N H Acid Amino H H O
R H O R R H O H H O H H N C C C H2N C O¯H O H O H O H C C N C N N H H C C C O R O O R Peptide Bond Peptide Bond Peptide Bond R R H H O O N N C C C C C C H2N H2N C C O H O O R R Making a Polypeptide Polypeptide Growth Polypeptide production = Condensation Reaction (dehyrdation synthesis)
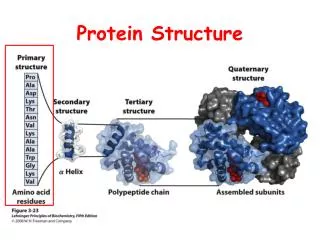
Why Investigate Protein Structure? Proteins are complex molecules whose structure can be discussed in terms of: primary structure secondary structure tertiary structure quaternary structure The structure of proteins is important as the shape of a protein allows it to perform its particular role or function
Protein Primary Structure The primary structure is the sequence of amino acids that are linked together. The linear structure is called a polypeptide http://www.mywiseowl.com/articles/Image:Protein-primary-structure.png
Protein Secondary Structure The secondary structure of proteins consists of: alpha helices beta sheets Random coils – usually form the binding and active sites of proteins Source: http://www.rothamsted.bbsrc.ac.uk/notebook/courses/guide/prot.htm#I
Protein Tertiary Structure Involves the way the random coils, alpha helices and beta sheets fold in respect to each other. This shape is held in place by bonds such as • weak Hydrogen bonds between amino acids that lie close to each other, • strong ionic bonds between R groups with positive and negative charges, and • disulfide bridges (strong covalent S-S bonds) Amino acids that were distant in the primary structure may now become very close to each other after the folding has taken place The subunit of a more complex protein has now been formed. It may be globular or fibrous. It now has its functional shape or conformation. Source: io.uwinnipeg.ca/~simmons/ cm1503/proteins.htm
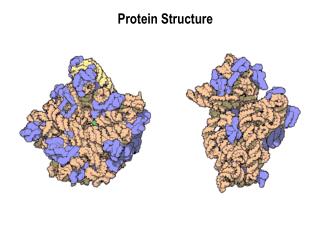
Protein Quaternary Structure This is packing of the protein subunits to form the final protein complex. For example, the human hemoglobin molecule is a tetramer made up of two alpha and two beta polypeptide chains (right) Source: www.ibri.org/Books/ Pun_Evolution/Chapter2/2.6.htm This is also when the protein associates with non-proteic groups. For example, carbohydrates can be added to form a glycoprotein Source: www.cem.msu.edu/~parrill/movies/neuram.GIF