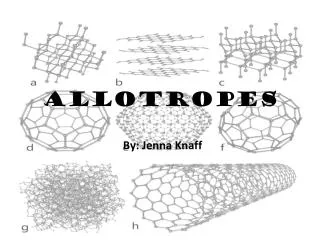
Allotropes
Allotropes. Li Jiawang. general knowledge about allotropes.

Allotropes
E N D
Presentation Transcript
Allotropes Li Jiawang
general knowledge about allotropes. • Allotropes are different forms of the same element with their atoms arranged differently from one another. They exhibit different behavior and characteristics. The conditions that changes allotropic forms are pressure, temperature and light. For example, for carbon to turn into diamond, we have to heat graphite to a very high temperature under great pressure . It is to simulate the conditions 140-190 kilometers down in the Earth’s mantle where diamonds are formed.
Arrangement of atoms in different forms of allotropes. Atomic arrangement in diamond Atomic arrangement in carbon Therefore, it can be concluded that when the allotropic forms are changed, the arrangement of atoms change as well, allowing the allotrope to exhibit different characteristics. Also, the number of atoms in a molecule also changes when change in allotropic forms take place.
In most circumstances, one allotrope will be more abundant than other allotropes. Examples are the O2 of oxygen which is far more abundant than the O3 of oxygen, ozone.
Let’s watch a video on allotropes of carbon. • http://www.youtube.com/watch?v=vYkyUqUa6vU
Summary of learning points in the video. • Structure of atoms in some allotropes of carbon, namely diamond, lonsdaleite, graphite, fullerenes. • Interesting fact about diamond: the diamond is not made up of molecules but is one big molecule itself. • There are different types of fullerenes, example C180 and C60.