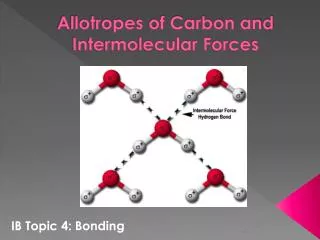
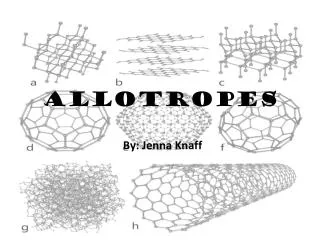
Allotropes
Allotropes. Casey Repasy Per. 6. Allotropes are…. Two or more forms of an element that have distinctively different physical or chemical properties. Ex: color of a diamond, or the arrangement of atoms in Phosphorous. . Different colored stones of a common carbon allotrope: diamonds. .

Allotropes
E N D
Presentation Transcript
Allotropes Casey Repasy Per. 6
Allotropes are… Two or more forms of an element that have distinctively different physical or chemical properties. Ex: color of a diamond, or the arrangement of atoms in Phosphorous. Different colored stones of a common carbon allotrope: diamonds.
Examples of Common Allotropes Carbon Allotropes (diamonds, graphite, nanotubes, fullerenes, glassy carbon, etc.) Phosphorous Allotropes (linked in chains or 2 dimensional hexagons) Oxygen Allotropes (regular oxygen and ozone) Sulfur Different arrangements of Phosphorus allotropes.
Uses or Applications of Different Forms of Allotropes It mostly depends on what kind of allotrope you have, but… Just think of a diamond, a carbon allotrope, and all of its uses: jewelry, tools or machinery, etc… Another carbon allotrope, glassy carbon, would be used by scientists in electrochemistry.
So, Why Does This Matter? Well, it relates back to mining…deciding on whether or not to mine a specific type of allotropes (like diamonds!) or not. We also just watched a movie on diamonds… We have been using the physical property aspect of this since our first lab!
And Now…To Review An allotrope is 2 or more forms of an ________ that have distinctively different ________ or _______ properties. What are some examples of carbon allotropes?
Sources Chemistry in the Community (our textbook) www.wisegeek.com/what-is-an-allotrope/htm www.chemistrydaily.com/chemistry/allotropesofcarbon