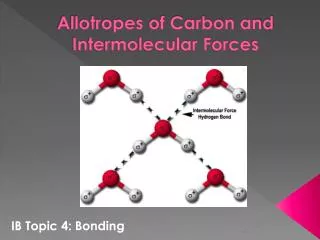
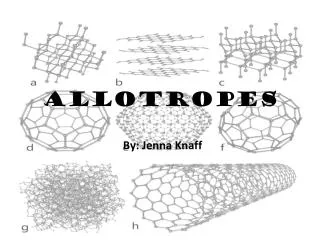
ALLOTROPES
ALLOTROPES. Olivia Corby Period 1 Fundamentals of Chemistry. DEFINITION. one of two or more existing forms of an element. Periodic Table of the Elements. Source : http://dictionary.reference.com/browse/allotrope. ALLOTROPES OF CARBON. d iamonds graphite.

ALLOTROPES
E N D
Presentation Transcript
ALLOTROPES Olivia Corby Period 1 Fundamentals of Chemistry
DEFINITION • one of two or more existing forms of an element Periodic Table of the Elements Source : http://dictionary.reference.com/browse/allotrope
ALLOTROPES OF CARBON • diamonds • graphite http://en.wikipedia.org/wiki/Allotropes_of_carbon
ALLOTROPES OF OXYGEN • dioxygen • ozone Diagram of the layers of the earth-specifically ozone layers http://en.wikipedia.org/wiki/Allotropes_of_oxygen
ALLOTROPES OF IRON • stainless steel • BBC THREE TYPES OF IRON ALLOTROPES: -alpha -gamma -delta http://en.wikipedia.org/wiki/Allotropes_of_iron
USES/APPLICATIONS • Diamonds-jewelry (rings, bracelets, necklaces, earrings, etc.) • Graphite-pencils • Stainless steel-buildings, silverware, pipes, etc.
HOW DOES THIS RELATE TO CLASS? • Balancing chemical equations-the product is a result of a compound which produces a brand-new substance • In order to create allotropes, substances sometimes need to be heated up or cooled down which is an example of a physical property • Because we have been studying the Periodic Table and understand how some of the elements are grouped, we can make connections to how some allotropes are formed
REVIEW QUESTIONS • What are 2 uses for any allotropes? • Ozone is an example of what kind of element allotrope?