B Cell Receptor Complexes
B Cell Receptor Complexes. SIGMA-ALDRICH. B Cell Receptor Complexes

B Cell Receptor Complexes
E N D
Presentation Transcript
B Cell Receptor Complexes SIGMA-ALDRICH
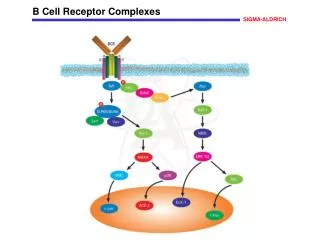
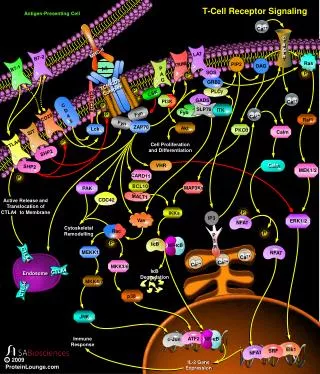
B Cell Receptor Complexes Reversible tyrosine phosphorylation plays an important role in regulating B cell function. In particular, binding of antigen to the B cell receptor (BCR) promotes the activation of several protein tyrosine kinases that, in conjunction with protein tyrosine phosphatases, alter the homeostasis of reversible tyrosine phosphorylation in the resting B cell. Members of the Src family are activated initially and phosphorylate CD79 and CD79ß, thereby creating phosphotyrosine motifs that recruit downstream signaling molecules. In particular, phosphorylation of the BCR complex leads to the recruitment and activation of the protein tyrosine kinase Syk, which, in turn, promotes the phosphorylation of PLC, Shc and Vav. Additionally, the Tec family member Btk is recruited to the plasma membrane where it is involved in activation of PLC . The SLP-65/BLNK adapter protein has recently been shown to play a role in BCR-induced recruitment and activation of key signal transducing effector proteins. Downstream intermediate signaling proteins are also activated including Ras which ultimately leads to the activation of MAP kinases including ERK, JNK and p38. References Kurosaki, T., et al., Regulation of the phospholipase C-2 pathway in B cells. Immunol. Rev., 176, 19-29 (2000). Pao, L.I., et al., Asymmetrical phosphorylation and function of immunoreceptor tyrosine-based activation motif tyrosines in B cell antigen receptor signal transduction. J. Immunol., 160, 3305-3314 (1998). Tan, J.E., et al., The adaptor protein BLNK is required for B cell antigen receptor-induced activation of nuclear factor-B and cell cycle entry and survival of B lymphocytes. J. Biol. Chem., 276, 20055-20063 (2001).