Chapter 14 – The Atmosphere
Chapter 14 – The Atmosphere The modern atmosphere Two most abundant gases: 78% N 2 21% O 2 Less abundant gases (< 1%) Argon Water vapor CO 2 (only about .035%) Non-gaseous components water droplets dust, pollen, soot and other particulates Fig. 17.6, p.437

Chapter 14 – The Atmosphere
E N D
Presentation Transcript
The modern atmosphere • Two most abundant gases: • 78% N2 • 21% O2 • Less abundant gases (< 1%) • Argon • Water vapor • CO2 (only about .035%) • Non-gaseous components • water droplets • dust, pollen, soot and other particulates
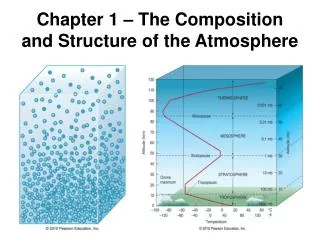
Thermal Structure of Atmosphere: Lower Layer • Troposphere – • to about 12 km (40,000 ft) elevation • All clouds & water vapor and most weather • Temperature decreases as elevation increases, because chief source of heat is radiated heat from the earth’s surface • Environmental Lapse rate: 6.4°C/1000m
Thermal Structure of Atmosphere: Upper Layers • Stratosphere – heated primarily by solar radiation • Ozone (O3) layer absorbs UV energy, causing temperatures to rise • Above 55km (stratopause) temps fall again • Mesosphere – thin air (can’t absorb energy), very cold up to 80km • Thermosphere – above 80km, temps rise rapidly (to just below freezing!)
Latent Heat • Sometimes heat transfer causes a change of state – change from solid to liquid, or liquid to gas. This occurs without a change in temperature. Examples: • Water stores energy when it changes from a liquid to a gas, since it absorbs the energy, but does not change temperature. • Water releases energy when it changes from a gas to a liquid. This is the source of wind energy found in hurricanes.
Latent Heat • Latent heat – energy stored or released during a phase change without temperature change
Humidity H2O in air exists in 2 forms: vapor (invisible) and liquid e.g. rain, fog, clouds. Saturation: Maximum quantity H2O vapor that air can hold. Additional H2O condenses into liquid water. Saturation increases with temperature (hot air can hold more water vapor than cold air. Conditions left of curve cause water vapor to condense.
Relative Humidity Relative humidity (RH)- ratio of actual amount H2O vapor in air to the amount at saturation. Often, as the days warms up, the air, more water will evaporate into the air (actual amount), but since the temperature has increased, so has the amount of water needed for saturation Therefore, the RH stays about the same.
Relative Humidity What happens if the air now cools down? Actual amount of water vapor in the air stays the same but amount needed for saturation decreases. Therefore the RH increases.
Relative Humidity When the relative humidity reaches 100%, the air is saturated, regardless of actual amount of water vapor present. At low temperatures, 100% RH means less water vapor than at higher temperatures.
Dew Point Temperature Dew point temperature – temperature at which RH = 100 %. If saturated air cools below the dew point temperature, water vapor will condense into liquid. At the dew point temperature, clouds, fog rain or dew appear.
Solar Energy (Insolation) • Also called solar radiation, although NOT radioactive! • Composed of electromagnetic waves with different properties depending on wavelength, frequency • Longwave (low frequency): includes heat (infrared), radio waves • Shortwave (high frequency): includes visible light as well as ultraviolet, x rays, gamma rays • Electromagnetic spectrum – shows EM wavelengths by frequency and wavelength
Shortwave solar energy • The highest frequencies (xrays, gamma rays) are absorbed by oxygen atoms in the thermosphere and O2 gas in the mesosphere. • Ultraviolet (uv) absorbed by the ozone in the stratosphere. • Visible light waves (still considered shortwave) pass through and are absorbed by earth.
Ozone high and low The ozone layer in the stratosphere is a good thing because it protects life on Earth from harmful UV rays The Ozone in the troposphere is a bad thing because it damages hearts and lungs.
Depletion of the ozone layer Ozone – O3 forms in the stratosphere and absorbs UV energy, which can be harmful Halons and Chlorofluorocarbons (CFCs), – are organic compounds that destroy ozone in the ozone layer
Depletion of the ozone layer Ozone hole – reported in 1985 and linked to CFCs in Antarctic ice clouds Many nations of the world agreed to reduce or stop use of these CFCs and halons Most industrial countries no longer produce CFCs. Since banning CFCs, the hole may be decreasing
Absorption and Emission • Absorption of radiation – electrons of absorbing material are “excited” by increase in energy • Increase in temperature; physical/chemical change • Examples: sunburn, cancer • Emission of radiation – excited electrons return to original state; radiation emitted as light or heat • Example: earth emits radiation in the form of heat after absorption of solar energy (light)
Reflection and Albedo • Reflection–electromagnetic radiation bouncing of from a surface without absorption or emission, no change in • material • energy wavelength • Albedo – proportional reflectance of a surface • a perfect mirror would have an albedo of 100% • Glaciers & snowfields approach 80-90% • Clouds – 50-55% • Pavement and some buildings – only 10-15% • Ocean only 5%! Water absorbs energy.
the Radiation Balance • Sun emits EM radiation of all wavelengths, but primarily shortwave (i.e. light). • Earth’s surface absorbs this energy • Most is re-emitted upward, as heat (longwave) • Greenhouse Effect and Counterradiation • “greenhouse gases” (water vapor, carbon dioxide, methane) let shortwave energy pass, but absorb and longwave energy radiated upward by the Earth. • this longwave energy is re-radiated in all directions, some of it returning to the Earth’s surface.
Cloud Formation Clouds – made of water droplets and/or ice particles (not water vapor, which is invisible) Cloud particles grow around a tiny solid surface; these are called condensation nuclei (singular nucleus). Examples include dust, smoke, pollen, sea salt pollutant particulates.
Cloud Formation clouds form when the air is saturated (100% relative humidity) and H20 vapor condenses. air must cooled to dew point for saturation to occur. Large scale cooling occurs when an air mass is lifted to a higher level in the atmosphere, due to the Adiabatic Principle.
When a gas expands, it’s temperature decreases. When a gas is compressed, it’s temperature increases. The adiabatic process refers to a temperature change caused by a change in pressure. The Adiabatic Process
Adiabatic (expansional) cooling – occurs when rising air expands due to decrease in air pressure. Adiabatic (compressional) warming - sinking air compresses, due to increase in air pressure.
For clouds to form by adiabatic cooling, the air mass must rise to where the air pressure is less; then it can expand The Adiabatic Process
What makes air rise? Warm air is less dense (lighter) than cool air. Water vapor is less dense (lighter) than dry air. Air near earth’s surface warms, picks up water vapor from near the surface of the Earth, then rises. Adiabatic cooling lowers temperature to dew point, which causes cloud formation. Air may be “helped” to rise by various means.
Adiabatic Lapse Rates Dry: 10°C/1000m Wet: approx 5°C/ 1000m <--- lifting condensation level
Clouds Cloud families – based on height • Low: to 10,000 ft above the surface • Middle: 10,000-20,000 ft above the surface (prefix alto) • High – above 20,000 ft high (prefix cirro) • Clouds with vertical development Cloud classes - based on shape • stratiform – layered clouds • cumuliform - globular
Cirrus – high altitude clouds Formed from ice crystals, not liquid water drops. Strong winds at altitude blow them into wisps (“mare’s tails”). No rain (or snow) comes directly from them, but they are often an indication of unsettled weather in the near future.
Low Stratus Clouds Horizontally layered, sheet-like clouds. Form in relatively stable conditions when air stops rising as soon as condensation occurs. Causes a gray overcast sky that may persist for days. Nimbostratus clouds bring steady rain or snowfall (nimbo = rain).
Cumulus clouds display vertical development Fluffy, white clouds with flat bottoms and billowy tops that may rise as much as 4 miles up in the air. Form from relatively unstable air which continues to rise after condensation takes place. Flat base of cloud is where condensation started. Rain or snow comes in the form of brief, intense showers.
Other Cumulus Clouds Fair weather cumulus Cumulonimbus
Lenticular clouds form as moist air flows up and over a mountain peak. They are classified as altocumulus. Lenticular clouds indicate high winds aloft.
Fog Fog is a cloud that forms at or very close to the ground. The picture shows an example of radiation fog, from air near the earth’s surface which cooled by radiation during the night. Fog usually disappears during the day, when the sun warms the earth and it begins to emit heat.
San Diego Marine Layer The marine layer is an example of an advection fog: warm air over the ocean takes in water layer, but as prevailing west winds blow it over the land, the water vapor condenses
Precipitation • Clouds sometimes release moisture as precipitation • Occurs as rain, hail, snow or sleet • 3 mechanisms that cause air to rise, leading to cloud formation and precipitation • Orographic precipitation • Cyclonic precipitation • Convection-Convergence precipitation
Orographic Precipitation Prevailing winds push air up against, and then over, a mountain range This process is responsible for our deserts in eastern San Diego County
Rainfall Map Orographic precipitation occurs on west side as air is lifted over mountains. Great Central Valley has less rain than coastal regions. Death Valley and Mojave Deserts are rainshadow deserts.
Cyclonic Precipitation (Frontal Wedging) When moving warm and cold air masses meet (a “front”), more dense cold air forces the less dense warm air to rise. This happens when a storm (cyclone) passes through
Convection-Convergence Precipitation • Forms when warm, moist air • Heats up near a hot surface • Rises and cools adiabatically • Condenses and forms clouds • May form fair weather cumulus • If conditions are unstable, can lead to precipitation in the form of thunderstorms.