Warm-Up 11/13/12
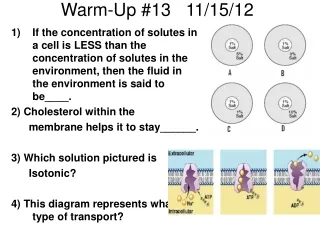
Warm-Up 11/13/12. COPY and ANSWER the following questions on p. 38 in your INB. (Use pgs 472-476 in your textbook for help) What is the difference between temperature and heat? What are the three temperature scales? What is the relationship between thermal energy and temperature?

Warm-Up 11/13/12
E N D
Presentation Transcript
Warm-Up 11/13/12 • COPY and ANSWER the following questions on p. 38 in your INB. (Use pgs 472-476 in your textbook for help) • What is the difference between temperature and heat? • What are the three temperature scales? • What is the relationship between thermal energy and temperature? • What is the relationship between thermal energy and heat?
In everyday life we used the words heat and temperature interchangebly • But, in physical science, while they are both related to thermal energy, heat, and temperature are not exactly the same.
Temperature • is defined as the average kinetic energy of the particles (atoms and molecules) in an object. • Thus, the higher the temperature the faster the molecules are moving • We observed this when we studied the motion of particles in a solid vs. a liquid vs. a gas. Particles in a gas have the greatest movement or kinetic energy.
Thermal Energy Relationships • a. As temperature increases, so does thermal energy (because the kinetic energy of the particles increased). • b. Even if the temperature doesn’t change, the thermal energy in a more massive substance is higher (because it is a total measure of energy).
Measuring Temperature • Temperature can be measured in degrees Fahrenheit or Celsius and Kelvin • All objects above absolute zero have thermal energy due to the motion of its particles • Absolute zero is an indication that there is no motion in an object’s particles
Heat • Is defined as the transfer of thermal energy from one object to another • So , heat is the flow of thermal energy • Heat always flows from a warmer object to a cooler object.
There are three ways in which heat transfer happens • Conduction – transfer of heat energy from one solid object to another when they are in direct contact. • Convection – transfer of heat energy through a flowing liquid or gas. • Radiation – transfer of heat energy in the form of electromagnetic waves. This is how heat energy can travel through the vacuum of space.
Conductors vs. Insulators • Materials that conduct heat easily are called Conductors. All metals are good conductors. • Materials that resist the flow of heat are called Insulators. Glass, plastic, and wood are examples of insulators.
Specific Heat Some things heat up or cool down faster than others.
Specific Heat • Specific heat is the amount of heat required to raise the temperature of 1 kg of a material by one degree (C or K). 1) C water = 4184 J / kg C 2) C sand = 664 J / kg C
Now, apply what you have learned about temperature, heat, and heat transfer by completing the appropriate sections of the “Thermal Energy” worksheet