Phase Equilibrium
Phase Equilibrium. A phase : a physically distinct portion of a system that is separated from the other portion by a boundary. A component : a chemically different constituents that participates in the formation of phases. Example :. Ice cubes. Two Phases One Component. water.


Phase Equilibrium
E N D
Presentation Transcript
Phase Equilibrium • A phase : a physically distinct portion of a system that is separated from the other portion by a boundary. • A component : a chemically different constituents that participates in the formation of phases.
Example: Ice cubes Two Phases One Component water
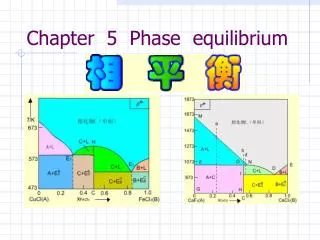
Gibbs’ Phase Rule • used to determine the degree of freedom which is the least number of independent variables, e.g.: temperature, pressure, and concentration affecting various phases at equilibrium. • F = C – P + 2 • Where; • F is the no. of degrees of freedom. • Cis the no. of components. • Pis the no. of phases exist in equilibrium.
Solids are soluble in liquids , while liquids are miscible in liquids. • Types of miscibility: miscible Partially miscible immiscible Ethyl alcohol /water phenol / water mercury / water
Factors affecting miscibility of phenol in water: 1- Concentration Water rich phase few drops of phenol few mls of phenol Phenol rich phase Water / phenol One phase water / phenol Two phases
Factors affecting miscibility of phenol in water: 2- Temperature Increase temperature Critical Soln. Temperature “upper consolute temp.”
Determination of critical solution temperature of phenol/ water system: Tie the ampoule with a thread If the difference between the temp. at which turbidity disappears and re-appears is > 2ºC, heat again and record the temp. at which turbidity disappears again. Suspend the amp. So that the contents of the ampoule must be completely immersed in the water bath Record the temperature at which the turbidity disappears (T1) using a thermometer In a beaker put water to form water bath Heat WB gradually with continuous shaking of the amp up & down Turn off the flame and record the temperature at which turbidity re-appears (T2)
Precautions : 1- Be very cautious with the ampoule (phenol injury is 3rd degree burn). 2- Suspended ampoule must not touch the wall or bottom of the beaker. 3- when recording the temperature, the suspended thermometer must not touch the wall or bottom of the beaker. 4- If the difference between the temp. at which turbidity disappears and re-appears is > 2ºC, heat again and record the temp. at which turbidity disappears again.
Exam 83%