Host-controlled restriction and modification system
Host-controlled restriction and modification system.

Host-controlled restriction and modification system
E N D
Presentation Transcript
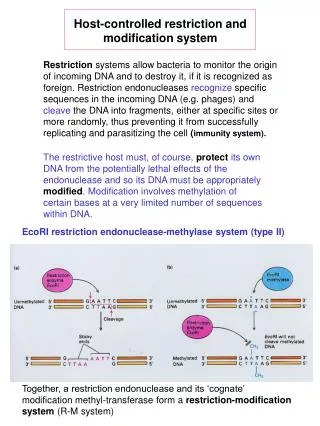
Host-controlled restriction and modification system Restriction systems allow bacteria to monitor the origin of incoming DNA and to destroy it, if it is recognized as foreign. Restriction endonucleases recognize specific sequences in the incoming DNA (e.g. phages) and cleave the DNA into fragments, either at specific sites or more randomly, thus preventing it from successfully replicating and parasitizing the cell (immunity system). The restrictive host must, of course, protect its own DNA from the potentially lethal effects of the endonuclease and so its DNA must be appropriately modified. Modification involves methylation of certain bases at a very limited number of sequences within DNA. EcoRI restriction endonuclease-methylase system (type II) Together, a restriction endonuclease and its ‘cognate’ modification methyl-transferase form a restriction-modification system (R-M system)
L’utilizzo degli enzimi di restrizione hanno permesso di realizzare prima molecola chimerica (Chimera creatura mitologica con la testa di un leone, il corpo di capra e la coda di serpente). 1973 - Stanley Cohen/Herbert Boyer (Stanford University California) Utilizzando il plasmide pSC101(contenente un sito unico per EcoRI, e la resistenza alla tetraciclina) riuscirono ad inserire DNA esogeno, proveniente dal plasmide p1258 di S. aureus (4 siti per EcoRI e Ampres) ligasi trasformazione di E.coli selezione per Ampres e Tetres .
The discovery of restriction and modification system The phenomenon of restriction and modification were well illustrated and studied by the behaviour of phage lambda on two E.coli strains (Aber & Dussoix 1960s). E.coli B Isolation of lambda E.coli B Efficiency of plating = 1 E.coli K Efficiency of plating = 10-4 Phages are said to be restricted by the second host strain ? Isolation of lambda E.coli K EOP = 1 E.coli B EOP= 10-4 Phages re-plated on E.coli K are no longer restricted. This non heritable change conferred upon the phage by the second host strain is called modification ?
The discovery of restriction and modification system (Type I) The restriction endonucleases Eco-K and Eco-B have been characterized in 1968. E.coli B Isolation of lambda E.coli B EOP= 1 E.coli K EOP = 10-4 Phages are restricted by the host Eco-K nuclease Eco-B nuclease does not cut DNA methylated in B-specific sites Isolation of lambda E.coli K EOP = 1 E.coli B EOP= 10-4 Phages re-plated on E.coli K are no longer restricted since Eco-K nuclease does not cut DNA methylated in K-specific sites Eco-B nuclease cuts DNA methylated in K-specific sites; few phages escape the hydrolysis and are modified in B-specific sites
Characteristics of restriction endonucleases Type III Type I Type II 1% ~ 98% 1%
Type II restriction enzymes Nearly 3000 restriction enzymes of type II have been found, exhibiting over 200 different specificities (about 200 enzymes are now purified and many of them are cloned and over-expressed in E. coli). Restriction enzymes are species non-specific: enzymes of the same specificity occur in different species (e.g BsaHI from B. stearothermophilus and AhaII from the blue-green alga Aphanothece halophytica). The genes for R.E. are often located on the chromosome, sometimes on plasmids and very occasionally located on prophages (it appears that the genes for these enzymes shuffle between microorganisms and that there is a natural selection for variety).
Type II (binary systems-separate endonuclease and methylase) This class of enzyme is particularly useful for gene manipulation, and present day DNA technology (since 70’, isolation of the first type II enzyme HindII from Haemophilus influenzae) is totally dependent upon our ability to cut DNA molecules at specific sites, using type II restriction enzymes. These enzymes recognize a particular target sequence (4 - 8 nucleotides) in a duplex DNA molecule and break the polynucleotide chains within, or near to, that sequence to give rise to discrete DNA fragments of defined length and sequence. They consist of a single polypeptide and require only Mg++ ions. Recognition sequences are symmetric, some sequences are continuous (e.g. G A T C), some are interrupted (e.g. G A N T C). Type II S (shifted cleavage) systems, like MboII or FokI, differ from standard type II system in having asymmetric recognition sequences. Cleavage occurs only on one side, at a point some distance away (fewer than 20 nucleotides). Two-fold axis of simmetry EcoRI Symmetrical, staggered cleavage of a short fragment of DNA by the type II restriction endonuclease EcoRI. The bold arrows show the sites of cleavage in the DNA backbone. (S=deoxyribose sugar; P=phosphate group)
OH O- P OH O O Single Stranded Nicks in DNA The cleavage reaction occurs on the backbone of one DNA strand Hydrolysis of this ester bond 3’ 5’ H2O Frequency of cutting of recognition enzymes Sau3A (GATC) cuts (¼)(¼)(¼)(¼) = once every 256 base pairs BamHI (GGATCC) cuts (¼)(¼)(¼)(¼)(¼)(¼) = once every ~4Kb HindII (GTPyPuAC) cuts (¼)(¼)(½)(½)(¼)(¼) = once every ~1Kb NotI (GCGGCCGC) cuts (¼)(¼)(¼)(¼)(¼)(¼)(¼)(¼) = once every ~ 65Kb (assuming G/C = A/T, which is often does not)
However eukaryotic DNA has a low content of CpG dinucleotides; the sequence recognized by HpaII (CCGG) is represented once in SV40 DNA (5.2 Kbp), but there are 26 sites in plasmid pBR322 (4.3 Kbp) Unit definition One Unit is defined as the amount of enzyme required to produce a complete digest of 1 g of DNA in 60 minutes in a reaction volume of 50 ml. “Sticky” ends (estremità protudenti) 5’ overhang (EcoRI) 5’-GAATTC-3’ 5’-G-OH PO4-AATTC-3’ 3’-CTTAAG-5’ 3’-CTTAA-PO4 HO-G-5’ + 3’ overhang (PstI) 5’-CTGCAG-3’ 5’-CTGCA-OH PO4-G-3’ 3’-GACGTC-5’ 3’-G-PO4 HO-ACGTC-5’ + “Blunt” ends (estremità piatte) 5’ overhang (SmaI) 5’-CCCGGG-3’ 5’-CCC-OH PO4-GGG-3’ 3’-GGGCCC-5’ 3’-GGG-PO4 HO-CCC-5’ +
BglII 5’ A-G-A-T-C-T T-C-T-A-G-A 5’ Sau3A 5’N-G-A-T-C-N N-C-T-A-G-N 5’ All these sticky ends are compatible BamHI 5’ G-G-A-T-C-C C-C-T-A-G-G 5’ Isoschizomers Most of the enzymes found today turn out to be duplicates (isoschizomers) of already discovered sequence specificities. Two or more different enzymes may recognize identical sites. (e.g. MboI also cleaves at GATC, and so is an isochizomer of Sau3A.) The three enzymes produce an identical protruding tetranucleotide sequence. Thus, for example, DNA fragments generated by complete with Sau3A can be subcloned in a vector that has been cleaved with BamHI. Is the BamHI site regenerated or is it lost?
Type II R.E. interact with sequences that are inverted repeats, and hence symmetric. Such sequences are said to be palindromic. 5’-GGATCC-3’ 3’-CCTAGG-5’ Bam H1 site: Most strains of E.coli contain two site specific DNA methylases: dam transfers a methyl group from SAM (S-adenosyl-methionine) to the N6 position of the adenine residue in the sequence 5’ G A T C 3’. This system distinguishes the strands of newly replicated DNA by methylating adenines. It is involved in control of replication and in marking DNA strands for repair. dcm methylates the internal cytosine in the sequence 5’ C C A G G 3’ or 5’ C C T G G 3’ Some R.E. will not cleave DNA methylated by either dam or dcm: ClaI GATCGATMboIGATC XbaI TCTAGATC ScrFI CCNGG StuI AGGCCTGG Other R.E. whose recognition sequence are identical to or overlap the dam or dcm methylase sites, are insensitive to DNA methylation: BamHI GGATCC BglII AGATCT BstNI CC(A/T)GGSau3AIGATC (methylated A or C in red)
Type II enzymes 3’-OH protruding; 8-bases recognition sequence (rare cutting enzyme) Blunt-end 5’-P protruding (sticky ends) 3’-OH protruding (sticky ends)
Activity of restriction endonucleases in different salt conditions • EnzymesActivity in different buffer (n.) • 1234 • BamHI + +++ +++ +++ • EcoRI ++ +++ +++ +++ • HindIII +++ +++ + +++ • SmaI (25°C) + + + +++ • SalI + + +++ ++ • XmaI +++ + + +++ • TaqI (65C°) ++ ++ ++ +++ • +++ 30-100 % of the optimal activity can be obtained in this buffer. • ++ 10-30 % of the optimal activity can be obtained in this buffer. • + <10 % of the optimal activity can be obtained in this buffer. • Buffer n. • 10 mM Bis Tris Propane-HCl, 10 mM MgCl2, 1 mM DTT (pH 7,0) • 10 mM Tris-HCl, 10 mM MgCl2, 50 mM NaCl, 1 mM DTT (pH 7,9) • 50 mM Tris-HCl, 10 mM MgCl2, 100 mM NaCl, 1 mM DTT (pH 7,9) • 20 mM Tris-acetate, 10 mM Mg-acetate, 50 mM K-acetate, 1 mM DTT (pH 7,9)
5’-C-G-G-T-A-C-T-A-G-OH 3’-G-C-C-A-T-G-A-T-C-T-T-A-A-PO4 Ligation of compatible sticky ends Human DNA cleaved with EcoRICorn DNA cleaved with EcoRI PO4-A-A-T-T-C-A-G-C-T-A-C-G-3’ HO-G-T-C-G-A-T-G-C-5’ + Complementary base pairing 5’-A-C-G-G-T-A-C-T-A-GA-A-T-T-C-A-G-C-T-A-C-G-3’ 3’-T-G-C-C-A-T-G-A-T-C-T-T-A-A G-T-C-G-A-T-G-C-5’ + DNA Ligase, + rATP 5’-A-C-G-G-T-A-C-T-A-G-A-A-T-T-C-A-G-C-T-A-C-G-3’ 3’-T-G-C-C-A-T-G-A-T-C-T-T-A-A-G-T-C-G-A-T-G-C-5’ recombinant DNA molecule
Restriction mapping of a linear DNA molecule EcoRI BamHI EcoRI + BamHI No enzyme 1650 1) There are two EcoRI sites. 2) There are two BamHI stes. 3) The 300 bp fragment is not cleavage by BamHI. 4) The 600 bp fragment is not cleavage by EcoRI. 5) The EcoRI 850 bp fragment yields the 600 bp and 250 bp fragments in the double digestion. 6) The BamHI 950 bp fragment yields the 400 bp, 300 bp and 250 bp fragments in the double digestion. 7) The EcoRI 500 bp fragment yields the 400 bp and 100 bp fragments in the double digestion.
300 600 E B E 600 250 E 850 400 B B 950 100 E 500














![[Light Host Management System]](https://cdn2.slideserve.com/4530772/light-host-management-system-dt.jpg)





