Protein Structure
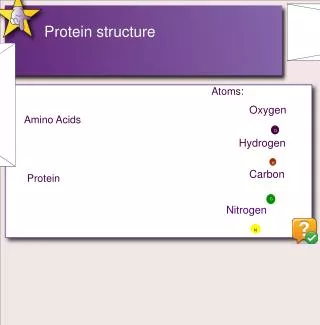
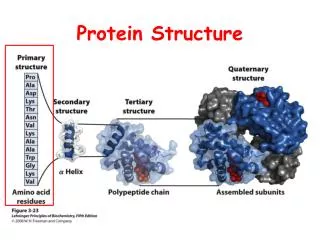
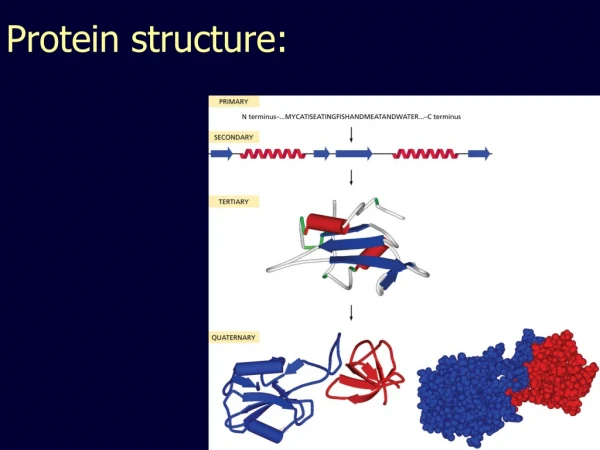
Protein Structure. Lecture 2/26/2003. Protein Structures. A study in the structure-function of proteins. Amino acid sequence dictates function. Structures are not “static” but breath and vibrate Protein dynamics (movement) can be linked to function. Globular proteins = enzymes and catalysts

Protein Structure
E N D
Presentation Transcript
Protein Structure Lecture 2/26/2003
Protein Structures A study in the structure-function of proteins. Amino acid sequence dictates function. Structures are not “static” but breath and vibrate Protein dynamics (movement) can be linked to function
Globular proteins = enzymes and catalysts Fibrous proteins = structural or connective role. Structure - function relationships Some residues and chains are just disordered “Floppy” flexible which maybe required for function
Fibrous (structural) proteins Keratin • Nails, hair, horns and feathers • or forms • 30 variants, tissue specific • type I and type II • acidic negative charge basic positive charge • keratin - • hair- 20 M diameter • macrofibril 2000 Å parallel to hair • microfibril 80 Å and high sulfur cement protein.
keratin proteins are helical but spacing differs from an a helix a 5.1 Å vs. 5.4 Å pitch. This change in pitch forms closely associated pairs of helices. Each pair consists of a type I and type II protein Left-handed coil coiled-coil 310 AA residues 7-residue pseudo repeat. Helical wheel - Look down an helix and residues stick out from center of helix 3.6 residues/turn 360 = 100 per residue 3.6 a - b - c - d - e - f - g a repeat on side of helix
Helical wheel diagram a and d residues are nonpolar. Protofilaments antiparallel strands
a - d are non-polar and face the same side of helix. 3.6 residues/turn 3.5 residues hydrophobic repeat The hydrophobic strip aligns between two helices with 18 inclination from one to another. They fit well together Dimer protofilament microfibril macrofibril hair keratin rich in cys and forms disulfides hard keratin cys content is high soft keratin cys content is cyst low Perms reduce R-S---S-R bonds to 2R-SH Curly hair has more Cys residues.
Protein helices are stretchy and can elongate When keratin is stretched it can form a more sheet like structure. b-keratin of feathers and nails are extended and have a more rigid and stiff consistency epidermolysis bullosa simplex and epidermolytic hyperkeratosis are keratin related diseases involved in the loss of mechanical integrity of the shin.
Silk Fibroin a b pleated sheet From spider and insect webs, cocoons, nests and egg sacks. An almost fully extended sheet that cannot stretch and is strong. This is why spiderman can support his weight on the web material!! Fibroin and sericin = web sericin is an amorphous gummy protein Adult moths dissolve (hydrolyze) their cocoons by cocoonase, this digests sericin, clothmoths do the same. Boiling water also removes sericin and leaves fibroin or silk.
Extended parallel sheets of (-Gly-Ser-Gly-Ala-Gly-Ala-)N Ala from one sheet interdigitates with Ala from another sheet Silks from different species have different interdigitating groups and have differing physical properties.
Silk fibers are strong when extended but cannot be stretched because of the fully extended sheet form of fibroin
Collagen - Triple helical cable Bones, teeth, cartilage, tendon, ligament, blood vessels and skin matrix Strong, flexible, stretchy Several types I [1 (I)]2 2I skin, bone tendon, cornea vessels II 1 (II)3 cartilage III [1 (III)]3 vessels, fetal skin Type I 285 kDA 14A wide 3000 A long 30 distinct peptide types 16 variants
1/3 Gly 15-30% -4-Hydroxyproline (Hyp) some 5-Hydroxylysyl (Hyl) 4-Hydroxyprolyl 3-Hydroxyproylyl (4-Hyp) (3-Hyp) C C N CH N CH 1 2 1 2 H 3 CH2 H3C 5 5 3 C H3C 4 OH 4 C C OH H H H
Gly-X-Y X often Pro Y often Hyp like a poly Gly or poly Pro helix Left-handed 3.0 residues/turn pitch 9.4 extended conformation the prolines avoid each other. 3 left handed helices combine in a triple rt handed coil.
Rope twist or metal cable longitudinal force (pulling) is supported by lateral compression opposite twisted strands prevents twists from pulling out.
Collagen helices are organized into fibrils. 689 A hole repeat 100 - 2000 A diameter different types make different arrays dark us light areas on fibril Hydrophobic repulsion drives fibril formation possible Van der Waals attraction due to packing. Collagen is 0.4 12% carbohydrate (linked sugars)
Vitamin C is required for hydroxyproline formation Hydroxyproline gives collagen stability and strength by H-bonding. Without prolyl hydroxylase collagen denatures at 24C instead of 39 to form gelatin. Scurvy-skin lesions, broken blood vessels, wounds don’t heal, teeth fall out, one cannot stand.