Vibrio Cholera
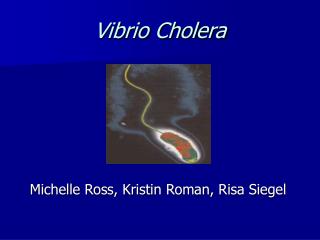
Vibrio Cholera. Michelle Ross, Kristin Roman, Risa Siegel. Vibrio Cholera. Micro and Molecular biology. Vibrio Cholera. Gram-negative Curved rod .5-.8 μ m width 1.4-2.6 μ m length Facultative anaerobe Single polar flagellum Chemoorganotroph Optimal growth 20-30 degrees. V. Cholera.

Vibrio Cholera
E N D
Presentation Transcript
Vibrio Cholera Michelle Ross, Kristin Roman, Risa Siegel
Vibrio Cholera Micro and Molecular biology
Vibrio Cholera • Gram-negative • Curved rod • .5-.8 μm width • 1.4-2.6 μm length • Facultative anaerobe • Single polar flagellum • Chemoorganotroph • Optimal growth 20-30 degrees
V. Cholera Gram-negative • lipopolysaccharide coat which provides protection against hydrophobic compounds • provides a surface for immune recognition
Divisions of V. Cholera • Biotype (biovar) different strains of the same bacterial species distinguished by a group of phenotypic or genetic traits • Serogroup bacteria of the same species with different antigenic determinants on the cell surface V. Cholera has more than 150 different serogroups, only two of which cause epidemic disease
V. Cholera01 serogroup Classic genome: 3.2-3.6 Mb El Tor (El) genome: 4 Mb 01 antigen is divided into 3 types: A,B,C • A antigen made of 3-deoxy-L-glycerotetronic acid • B, C antigen not been characterized
Horizontal Gene Transfer 1. acquisition of VPI 2. lysogenic conversion by phage 3. exchange of genes leads to expression of O-antigen and capsule
the 01 strain and the recent 0139 strain have different antigens expressed in the polysaccharide capsule the change in structure is thought to have arisen from a recombination event. V. Cholera
V. Cholera two circular chromosomes Chromosome 1 is larger (2.96 million base pairs) and carries many genes for essential cell functions and housekeeping Chromosome 2 is smaller (about 1.07 million base pairs and carries the integron island
Chromosome 1 • carries many genes for essential cell functions and housekeeping. It also contains important virulence genes, most of which have been acquired by lateral gene transfer from other species • chromosome one carries two bacteriophages. ONE VIRUS is called the V. cholera pathogenicity island phage (VPI), which infects and inserts its DNA into the bacterial chromosome and allows the synthesis of a pilus which the bacteria uses to attach to the host intestine SECOND VIRUS is called the cholera-toxin phage (CTX). The CTX phage inserts itself into chromosome one and the bacterium is then capable of secreting a powerful enterotoxin
Chromosome 2 The integron region is often found on plasmids and serves as a "gene capture system." This region may contain antibiotic resistance genes.
Pathogenesis of V. Cholera Cholera disease begins with ingestion of contaminated water or food. The bacteria that survive the acidic conditions of the stomach colonize in the small intestine. The cholera toxin (CT) is responsible for the severe diarrhea characteristic of the disease. Cholera Toxin CT is a proteinaceous enterotoxin secreted by V. Cholera
Cholera Toxin Structure • Composed of a AB subunit. The B subunit forms a pentameric “doughnut” like structure that binds the CT to the receptor on the eukaryotic cells Pathway • The A subunit contains the enzymatically active portion or the toxin • Proteolytic cleavage of the A subunit results in A1 and A2 peptide units which remain linked by a disulfide bond • Once the A subunit is internalized by the eukaryotic cell, the disulfide bond is reduced
Pathway continued The A1 subunit contains a ADP-ribosyltransferase which covalently modifies the G protein, which regulates adenylate cyclase. Adenylate cyclase mediates the formation of cAMP The increase in cAMP levels bring about the secretion of chloride and bicarbonate from the mucosal cells into the intestinal lumen The change in ion concentrations leads to the secretion of large amounts of water into the lumen, known as diarrhea
Genetics of Cholera Toxin Genes encoding CT ctxAB - recognized to be the genome of a filamentous phage CTXΦ (ctxA and ctxB) Transcription of ctxAB is regulated by several proteins CTXΦ genome can integrate into the host genome at a specific site, attRS The CTX genetic element also has a “core” region carrying several phage morphogenesis genes These entire CTX gene set is flanked repeated sequences, the attRS1 site The entire genetic element is 6.9kb The receptor for CTXΦ is Toxin-Coregulated Pili (TCP)
Toxin-Coregulated Pili Efficient colonization of V. cholera in the small intestine requires the expression of TCP’s • TCP’s are expressed on the surface of V. cholera • TCP’s are long laterally associated filaments The major pilin subunit is TcpA Genes for TCP production are clustered on the pathogenicity island located on chromosome 2
Regulation of Virulence Factors Expression of CT & TCP have been shown in vitro to be strongly influenced by changes in cultural conditions ie. temperature, pH, osmolarity, & growth medium composition CT & TCP are regulated via a cascade in which ToxR and TcpP control expression of ToxT, which is a transcriptional activator directly controlling several virulence genes ToxR & TcpP are inner-membrane proteins which interact with other transmembrane regulatory proteins ToxR/S proteins are required for transcription of toxT gene and are also important for ctx transcription and regulation other outer membrane proteins
Recap of phage movement V. cholerae did not always cause disease. Infection with the CTX phage gives the bacterium its toxinogenicity. The phage recognizes a pilus on the surface of the bacterium and uses it to enter the cell. Once inside the cell, the CTX phage integrates into the chromosome and the lysogen expresses cholera toxin. The CTX phage has received special attention because it is the first filamentous phage found to transfer toxin genes to its host. The important lesson from this discovery is that many different types of phage may carry virulence factors, and transfer of virulence genes by phage may be a major mechanism of evolution of new bacterial diseases.