Lewis Structures and Metallic Bonding
150 likes | 374 Views
Tuesday 10/19/10. Lewis Structures and Metallic Bonding. Agenda. Return Naming Compounds Quiz Collect Symbol Me Projects Lewis structures Metallic Bonding Hydrogen Bonding Jeopardy Review. Lewis structures. Atoms form bonds to gain a full shell of valence electrons. (Octet Rule)

Lewis Structures and Metallic Bonding
E N D
Presentation Transcript
Tuesday 10/19/10 Lewis Structures and Metallic Bonding
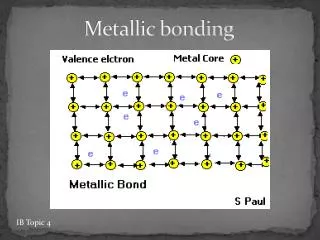
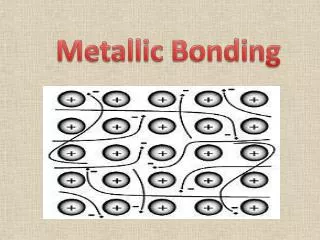
Agenda • Return Naming Compounds Quiz • Collect Symbol Me Projects • Lewis structures • Metallic Bonding • Hydrogen Bonding • Jeopardy Review
Lewis structures • Atoms form bonds to gain a full shell of valence electrons. (Octet Rule) • If electrons are not involved in bonding, then they are lone pairs. • Each bond is made up of 2 electrons.
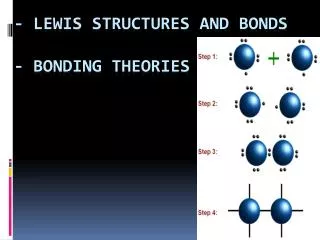
Steps • Count up the total # of valence electrons. If it’s an ion, then add or subtract e- as needed. • Connect all the atoms with single bonds. • Place the remaining electrons as lone pairs around the atom. • Create double or triple bonds if there are not enough electrons to go around. • ALWAYS FOLLOW THE OCTET RULE!!!!
#1 • Draw the Lewis structure for NO3-
#2 • Draw the Lewis structure for CCl3+
#3 • Draw the Lewis structure for CO2
#4 • Draw the Lewis structure for N2O4
#5 • Draw the Lewis structure for C2N24-
#6 • Draw the Lewis structures for PO43-
#7 • Draw the Lewis structure for OCN-
http://www.youtube.com/watch?v=lkl5cbfqFRM&feature=related\ • http://www.youtube.com/watch?v=LGwyBeuVjhU