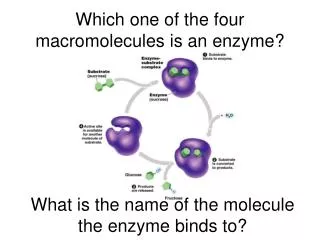
How do cells regulate enzyme activity?
Lecture 4: Strategies of enzyme regulation: covalent modification, feedback inhibition, and allosteric regulation. How do cells regulate enzyme activity?.

How do cells regulate enzyme activity?
E N D
Presentation Transcript
Lecture 4: Strategies of enzyme regulation: covalent modification, feedback inhibition, and allosteric regulation
How do cells regulate enzyme activity? • Long-term regulation: Biological demands upon enzymes are highly variable - some enzymes function only in particular parts of the body, others only in particular parts of the cell, others still only during certain times in development. These sorts of differences are long-term in their nature and are usually regulated at the level of protein synthesis (discussed in the next module), localization, and degradation. • Short-term regulation: Enzymes must also be regulated rapidly and reversibly to respond to ever-changing demands placed upon cells. How enzymes respond to changing cellular conditions is a central feature of their functions as biological entities.
Covalent modification of enzymes • One way in which proteins can be modified is through covalent modification. This regulation can assume many forms: (1) Proteolysis - Proteins can be cleaved by other enzymes called “proteases”; this can eliminate the activity of the targeted protein or can activate it. An example of the latter is the subject of a later lecture. (2) Addition/removal of small-molecule modifications - Several amino acid side chains can be chemically modified by the addition or removal of non-protein compounds. This can either activate or inhibit (or have no affect on) enzyme function. In some cases these are reversible.
Typical protein modifications An example of N-glycosylation:
Feedback inhibition • Feedback inhibition is a common means of regulating activity of an enzyme or a biological pathway by its product. • Here are two generic biological pathways: • In the top example, the pathway is linear, and the final product “E” inhibits the first enzyme of the pathway. Why does this make sense? • In the bottom “branched” pathway, the final product of one of the pathways “Y” inhibits the first reaction unique to this its synthesis. Predict which reaction will be inhibited by “V”…
Feedback inhibition • Feedback inhibition is a common means of regulating activity of an enzyme or a biological pathway by its product.
Rationalizing feedback inhibition • Why do cell use feedback inhibition to regulate enzyme function? It is wasteful to run a biological reaction/pathway if the product is not needed! Many pathways require an input of energy from cellular stores (we’ll see an example today), so producing an already abundant product would be disadvantageous to the cell…
What about activation? • Since feedback inhibition is an effective way to regulate enzyme function it would also make sense that cells should be able to simulate enzyme function in the event that the product of a reaction/pathway is needed. • This can be more complex, as in the example that we will be going through in a moment, but it is analogous to feedback inhibition…chemical clues from the cell are used as a tool to modulate (in this case activate) enzyme function. Which reactions from before do you think would be stimulated in this way?
How is the regulation actually accomplished? • Feedback inhibition (and the related activation phenomenon) are often the products of allosteric regulation of enzyme activity. •In this type of regulation, binding sites distant from the enzyme active site (“allo” means “other”) affect the level of activity at the active site by subtle alterations in the active site structure. These effects are thus transmitted through the enzyme’s structure. • Most often, allosterically regulated enzymes are composed of multiple subunits some of which are regulatory and others of which are catalytic.
Aspartate transcarbamylase as a classic example of enzyme allostery • ATCase catalyzes the rate-limiting and first unique step in CTP synthesis. • Note that cellular energy stores (in the form of ATP’s high-energy bonds) are used up in the pathway. • Any guesses as to whether ATCase is regulated? Why?
Let’s look at the ATCase structure(s) • ATCase is comprised of 12 subunits (3 regulatory dimers and 2 catalytic trimers) • The enzyme does not exist in one static structure, but instead fluctuates between two different states, called the relaxed, or “R”, state and the tense, or “T”, state.
What do ATCase reaction kinetics look like? • Does this look normal to you? NO! It turns out that the two forms of ATCase have different catalytic properties: • The T state is inactive and the R state is active. • What would cause this odd kinetics plot shape? Cooperativity! • What is going on at K0.5? Is each enzyme half active?
Now, how is ATCase regulated? What do you predict would be the effect of excessive amounts of CTP on ATCase structural behavior and on its kinetics?
What about an allosteric activator? • In times when nutrients are abundant, it is wise for cells to divide. As such the DNA in the cell needs to be duplicated (as will be described in the next module) which requires nucleotides such as dCTP. • In such conditions, how might you activate ATCase to produce more CTP? Obviously, one way is that cellular stores of CTP will be consumed, so CTP inhibition will be at a minimum. But there is another, more active, mechanism as well…any guesses?














![[virtual] cells](https://cdn1.slideserve.com/3553683/slide1-dt.jpg)