Spectrin repeats
130 likes | 387 Views
Spectrin repeats. Protein Motifs Session FRCPath Preparation Course 10/12/09. FEBS Letters Volume 513, Issue 1, 20 February 2002. Spectrin repeats (SR). Large modular repeat elements found in protein members of the spectrin superfamily.

Spectrin repeats
E N D
Presentation Transcript
Spectrin repeats Protein Motifs Session FRCPath Preparation Course 10/12/09
FEBS Letters Volume 513, Issue 1, 20 February 2002 Spectrin repeats (SR) Large modular repeat elements found in protein members of the spectrin superfamily Consist of 3 helices (A, B, C) that are separated by two loop regions (AB, BC) and arranged in a left handed triple helical bundle. Approx. 100 amino acids in each repeat Low sequence identity between repeats but all share similar 3D structure Common key conserved residues involved in the hydrophobic and electrostatic interactions that stabilise the bundle - invariant Tryptophan residue at position 17 of Helix A - highly conserved charged residues in all helices - a nearly invariant Tryptophan in the middle of Helix C - a Leucine residue at carboxyl end of Helix C There is a sequence of a few residues linking each repeat to the next Thought to have originated through duplication events of the exons coding the original progenitor SR Variation between repeats is suggestive of specialised functions.
Spectrin superfamily: • Cytoskeletal proteins that are cross-linking actin or linking actin to the cell membrane. • Examples include: -actinin, spectrin, dystrophin, utrophin, nesprins • Common features: • Multiple isoforms • Typical architecture • N terminal actin binding domain • Intervening variable number of SR and other functional motifs • C terminal region containing a distinct functional domain SR are also found in unrelated proteins involved in signalling pathways Examples include kalirin, MCF2 (dbl-protooncogene), PLEKHG4 (puratrophin-1)
SR function Structural role in assembly of complex proteins involved in cytoskeletal and signal transduction complexes - maintenance and modelling of cytoskeleton and organelles - organisation of networks of multiprotein interactions Mechanical properties – Elastic, flexible, dynamic - important in cytoskeletal proteins that are exposed to great mechanical stress Binding sites for interaction with other proteins
www.bioscience.org/2005/v10/af/1759/fig2.jpg Spectrin Cytoskeletal protein found in many cell types. Present in both vertebrates and invertebrates. Links plasma membrane to the actin cytoskeleton 7 genes in humans: - subunit : SPTA1 (erythrocyte isoform), SPTAN1 - subunit : SPTB (erythrocyte isoform), SPTBN1, SPTBN2, SPTBN4, SPTBN5 Composed of two antiparallel dimers of - and - subunits (tetramer formation) - and - subunits assembled in dimers in a head to tail mode Dimers arranged in a tetramer in a head to head manner • Scaffolding role: • Determination of cell shape • Arrangement of transmebrane proteins • Organisation of organelles (e.g. Golgi apparatus) • Involvement in DNA repair (- SpecIII)
http://www.ncbi.nlm.nih.gov/bookshelf/br.fcgi?book=mboc4&part=A1861http://www.ncbi.nlm.nih.gov/bookshelf/br.fcgi?book=mboc4&part=A1861 Red blood cell - spectrin based cytoskeleton Triangulated 2D meshwork composed of spectrin tetramers that are crosslinked by actin in association with accessory proteins. • The biconcave shape of red blood cells (RBCs) provides them with strength and flexibility for surviving stress during circulation and passing through narrow capillaries.
Spectrin in disease (1) • Mutations in spectrin and associated proteins results in disruption of the 2D meshwork causing abnormal RBCs membrane formation. • The abnormal shape causes RBC to become fragile and prematurely lost from circulation causing haemolytic anaemia. Hereditary Spherocytosis (HS): Spherical-shaped RBC (spherocytes) on the peripheral blood smear. Anaemia, jaundice, and splenomegaly, with variable severity. Common complications include cholelithiasis, hemolytic episodes, and aplastic crises. Recessive Hereditary Elliptocytosis (HE):Oval-shaped RBC Variable degree of haemolytic anaemia. Dominant Hereditaty pyropoikilocytosis (HPP): microspehrocytosis, poikilocytosis and thermal sensitivity of RBC. Severe presentation in infancy progression to elliptocytosis in later life. Recessive • Mutations detected in both alpha and beta spectrin subunits (HS, HE, HPP) • Found in the tetramerisation sites, linker sequence or spectrin-ankyrin interaction sites
Spectrin in disease (2) • SCA5 • Originally mapped in 11q13 in some families with cerebellar cortical atrophy, Purkinje cell loss and thinning of the molecular layer. • Mutations detected in SPTBN2 (III spectrin) • Regulates the glutamate signalling pathway by stabilizing the glutamate transporter (EAAT4) at the surface of the plasma membrane. • 2 in frame deletions within SR and 1 in CH domain reported • Mutations detected are likely to disrupt the binding to the actin cytoskeleton and disrupt the stabilisation of the membrane proteins. The loss of EAAT4 signalling over time could cause the Purkinje cell death observed.
Molecular Therapy (2006) 13, 241–249 Dystrophin • N terminal actin binding domain • 24 internal spectrin repeats forming a flexible and elastic rod domain • An internal actin domain (SR 11-17) • Cysteine rich domain • C terminal domain • The Cysteine rich domain and the C terminus domain interact with integral membrane proteins (dystrophin-associated glycoprotein complex) Dystrophin provides a structural and signalling link between the extracellular matrix and the intracellular actin cytoskeleton
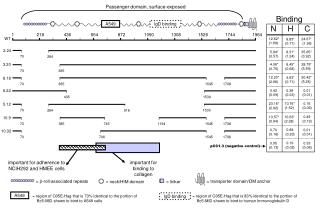
Dystrophin role: • Stability during muscle contraction • Force transduction; may link the contractile force produced in the intracellular domain to the extracellular environment • Maintaining membrane integrity • - Deficiency of dystrophin causes disruption of the dystrophin-glycoprotein complex. • Cell membrane becomes more fragile->damaged during muscle contraction • Complex cascade process leading to muscle fibres undergoing necrosis In DMD/BMD, partial deletions and duplications cluster in two recombination hot spots, one proximal at the 5' end of the gene exons 2-20 (30%), and one more distal exons 45-53 (70%) - DMD: 60% deletions, 5% duplication, remaining point mutations - BMD: 80% deletion, 10% duplications, remaining point mutations DMD: Mutations that lead to lack of dystrophin expression BMD: Mutations that lead to abnormal quantity or quality
Nesprins: family of nuclear associated proteins • Nesprin 1(SYNE1) and Nesprin 2(SYNE 2) • Widely expressed in skeletal cardiac and vascular smooth muscle • Multiple isoforms • N terminal actin binding domain, spectrin repeat rod domain, KASH domain C terminal domain (mediates nuclear membrane localisation) • Present in multiple subcellular locations including the nucleus, INM, ONM, organelles (e.g. mitochondria, Golgi), plasma membrane • Form a subcellular network that potentially links the plasma membrane to the nucleus, and disruptions of this network are likely to impair subcellular structure and function.
Nesprins in disease • Mutations detected in SYNE1 leading to premature termination of protein have been associated with ARCA1 or recessive ataxia of Beauce. • Late onset cerebellar ataxia, dysarthia, brisk lower limb reflexes, limb ataxia • Mouse models have shown that syne-1 highly expressed in Purkinje cells; loss may lead to disruption of cerebellar architecture leading to the ARCA1 phenotype • Mutations in SYNE1 and SYNE2 have been detected in patients with EMD phenotype • Fibroblasts from these patients exhibited nuclear morphology defects attributed to the nesprin/emerin/lamin defective interaction. • SYNE1 has been implicated in a family with an autosomal recessive form of arthrogryposis multiplex congenita.
References: Speicher and Ursitti 1994. Conformation of a mammoth protein. Current Biology Vol 4 No 2 p:154. Djinovic-Carugo et al 2002. The spectrin repeat: a structural platform for cytoskeletal protein assemblies. FEBS Letters Volume 513, Issue 1, 20 February 2002 Young and Kothary 2005. Spectrin repeat proteins in the nucleus. BioEssays 27: 144-152 Stabach et al 2009. The structure of ankyrin-binding site of -spectrin reveals how tandem spectrin repeats generate unique ligand-binding properties. Blood Vol 113, No 22 p:5377 Pascual Castrenasa and Saraste 1997. Evolution of the spectrin repeat. BioEssays 19: 811-817 De Matteis and Morrow 1998. The role of ankyrin and spectrin in membrane transport and domain formation. Current Opinion in Cell biology 10: 542 Ahn and Kunkel 1993. The structural and functional diversity of dystrophin. Nature Genetics Vol 3 p:283