METALLIC BONDS
METALLIC BONDS. Characteristics of Metal. Good conductors Malleable, ductile The simplest crystalline solids Each metal contains just one kind of atom. Compact and orderly High melting points and boiling points. Shiny or lustrous. Uses of metal. Copper is used for electrical cables .

METALLIC BONDS
E N D
Presentation Transcript
Characteristics of Metal • Good conductors • Malleable, ductile • The simplest crystalline solids • Each metal contains just one kind of atom. • Compact and orderly • High melting points and boiling points. • Shiny or lustrous
Uses of metal • Copper is used for electrical cables
Continued • Iron and steel are used to construct bridges, buildings and motor cars
Aluminium is used to make saucepans and thin foil. • Gold and silver are are used to make jewellery which does not tarnish.
Alloys • Alloys : A mixture of two or more metalic elements that form a solid solution. • The aim of making alloys is to make them less brittle, harder, resistant to corrosion, or have a more desirable color and luster. • Ex) steel (iron and carbon), brass (copper and zinc) bronze (copper and tin)
Types of alloys • 1)Substitutional alloys: The atoms of metal are the same size. • 2)Interstitial alloys: The atoms of metal are different sizes
Continued • Electrons get delocalized. • Delocalization : Electrons absorbs energy→ get excited→ moves freely (said to be create an “electronic highway”)
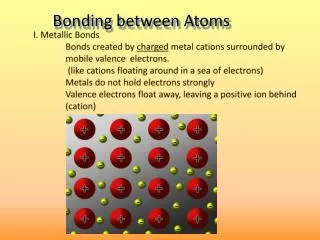
Metallic bond • Metallic bond: The force of attraction that holds metals together, it consists of the attraction of free floating valence electrons for positively charged metal ions.
Band theory • Delocalized electrons move freely through “bands” formed by overlapping molecular orbital. • These molecular orbital are so closely spaced on the energy scale that they are more appropriately described as a band.
Continued • Valence band:The closely spaced filled energy levels make up the valence band • Conduction band: The set of closely spaced empty levels is called the conduction band.
Example Copper Electron Configuration: 1s22s22p63s23p63d94s2 Conduction band Wall e- e- e- e- e- e- e- e- 4s2 3d9 3p6 3s2 2p6 2s2 1s2 e- e- e- e- e- e- Cu Cu Cu
Band Theory • The most characteristic property of any metal is high electrical conductivity. When an electron absorbs energy from an electric field, it is excited into an unutilized molecular orbital, constructed from all of the atomic orbitals in the entire metal. • As a result, this orbital is totally delocalized. Therefore, it provides a kind of electronic highway throughout the entire metal, allowing charge to be transported with great ease. • The band theory also explains why metals are ductile and malleable. Since the sea of delocalized electrons can immediately adjust to any deformation that occurs in the crystal structure, the strength and shape of the new (deformed) structure can be immediately restored.
Conductor vs Insulator • Conductor: Capable of conducting electric current • Ex) copper • Insulator: ineffective conductors of electricity • Ex) glass, wood
Semiconductors • They normally are not conductors, but will conduct electricity at elevated temperatures or when combined with a small amount of certain other elements • Ex) silicon, germanium (Group 4 elements)