Metallic Bond
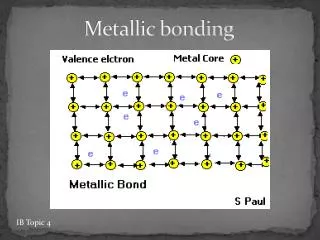
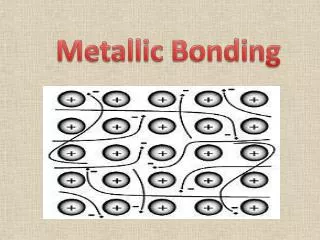
Metallic Bond. IB. The Periodic Table of Elements. Periodic Table. Formation of metallic bond. the metal atoms "lose" one or more of their outer electrons These electrons become delocalized, and free to move throughout the entire metal.

Metallic Bond
E N D
Presentation Transcript
Formation of metallic bond • the metal atoms "lose" one or more of their outer electrons • These electrons become delocalized, and free to move throughout the entire metal. • These negative delocalized electrons hold the metal cations together strongly.
Body Centered Cubic Chromium
Face-Centered Cubic Gold
Solids • Allmetals are solids at roomtemperatureexceptionmercury.
Properties of Metals Metals have luster. This means they are shiny
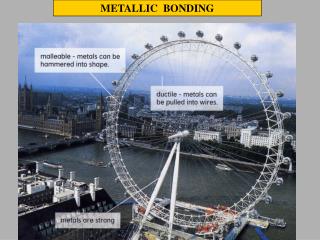
Properties of Metals Malleable
Properties of Metals Ductile metals can be drawn into wire.
Properties of Metals Metals have a high melting point. They are also very dense.
Properties of Metals Metals are good conductors of electricity and heat