Fats and Oils
220 likes | 1.47k Views
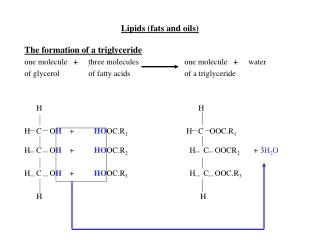
Fats and Oils. Fats and oils have the same basic structure. They are made in a condensation reaction when 3 fatty acid groups join with 1 molecule of glycerol. 3 water molecules are produced in each reaction. Glycerol.

Fats and Oils
E N D
Presentation Transcript
Fats and Oils • Fats and oils have the same basic structure. • They are made in a condensation reaction when 3 fatty acid groups join with 1 molecule of glycerol. • 3 water molecules are produced in each reaction.
Glycerol • Glycerol is a tri ol – it has 3 hydroxyl groups in its structure. A trihydric alkanol. • It’s chemical name is propan1,2,3 tri ol. CH2 – OH I CH2 – OH I CH2 - OH
Fatty Acids • Fatty acids contain a carboxyl functional group attached to a long chain. • The chain length can vary – even number of carbon atoms. • In fats the chains are saturated. • In oils the chains are unsaturated. O II HO – C – R ( R = carbon chain, C4 – C24) Usually C16 – C18
The Reaction GlycerolFatty Acids CH2 – OH + HOOC - R I CH2 – OH + HOOC – R* I CH2 – OH + HOOC – R**
The Product Fat or Oil CH2 – OCO – R I CH2 – OCO – R* + 3 H2O I CH2 – OCO – R** O II O – CEster link
The New Ester • The ester formed in the reaction is called a tri glyceride. • Fats and oils are mixtures of triglycerides. • The triglyceride can be broken down in a hydrolysis reaction to form glycerol and the 3 fatty acid molecules. • This is what happens to fat/oils when they are digested.
Uses of Fats and Oils • Fats and oils are used to produce energy. • They contain twice as much energy as proteins and carbohydrates. • We can compare this in the lab by burning equal masses of foods under a test tube of water and noting the overall T change. The higher the increase in T – the more energy released from the food.
Fats/Oils • Fats are grouped into: animal( beef, pork etc), vegetable( sunflower, olive etc.) and marine ( cod liver oil etc.) • Fats have a higher melting point – the fatty acid chains are saturated and so can be easily stacked together. More Van der Waals’ forces. • Oils have a lower MP – the fatty acid chains are unsaturated making stacking more difficult.
Hardening • We can harden or saturate oils by adding hydrogen to the fatty acid chain. • Hydrogenation • Hardened oils are used in margarine.
Soap • Soaps are made by the hydrolysis of fats and oils using any alkali. • The salt of the alkali is produced as well as glycerol. O II • R – C – O - Na +