Chapter 6
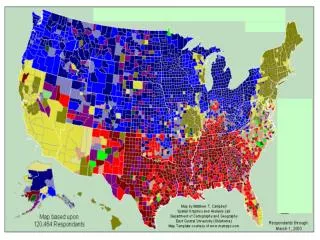
Chapter 6. The Periodic Table and Periodic Law. Remember: The Periodic Table is Your Friend!!. The Periodic Table. Periodic – means repeating pattern. 6 .1 History of the Periodic Table. Mendeleev: Arranged the periodic table according to atomic mass

Chapter 6
E N D
Presentation Transcript
Chapter 6 The Periodic Table and Periodic Law
Remember: The Periodic Table is Your Friend!! The Periodic Table • Periodic – means repeating pattern
6.1 History of the Periodic Table • Mendeleev: • Arranged the periodic table according to atomic mass • Similar chemical properties appeared in repeating intervals • Page 175 – Mendeleev’s Periodic Table • Only 63 elements were known at this time • Gaps were left in the table but Mendeleev predicted that these elements did exist
6.1 History of the Periodic Table • Moseley: • Arranged the periodic table in order of increasing atomic number • Question: Can you find at least 2 places on Mosely’s periodic table that would be different if Mendeleev was right? (That is if the periodic table was organized by atomic mass not atomic number) • Te and I, Co and Ni, Sg and Bh, Th and Pu, U and Np
6.1 History of the Periodic Table • Periodic Law: When elements are arranged in order of increasing atomic number there is a periodic pattern in their chemical and physical properties
Modern Periodic table • Groups: vertical columns (1-18) • Periods: horizontal rows (1-7) • Arrangement: by increasing atomic number with groups containing similar chemical and physical properties
5.2 Periodic Table and Electron Configuration • The periodic table consists of 4 different “blocks” • s – block • p – block • d – block • f – block • Pg 183 – Figure 6.8 shows the blocks
S - Block • Groups: 1 and 2 plus H and He • Group 1 – Alkali Metals • Extremely reactive, seldom found free in nature • Silvery, soft metal • Outer energy level = s1 (H is 1s1) • Group 2 – Alkaline Earth Metals • Reactive, seldom found free in nature • Harder and stronger than Group 1 metals • Outer energy level = s2 (He is 1s2)
P - Block • Groups: 13 – 18 except He • Group 17 – Halogens (Diatomic X2) • Most reactive nonmetals • React with metals to form “salts” • Outer energy level = s2p5 • Group 18 – Noble Gases • Unreactive, most stable elements • Outer energy level = s2p6
D - Block • Groups: 3-12 • Transition metals • High melting points, good conductors • Used for jewelry, power lines • Less reactive than Group 1 and 2 metals • Outer energy level: s and d have electrons
F – Block • Bottom Two Rows: Inner Transition Metals • Lanthanide – • Shiny metals • Used in TV sets to emit color • Outer energy level: 6s and 4f • Actinides – • Mostly manmade and radioactive • Used for nuclear fuel, smoke detectors, A – bomb • Outer energy level: 7s and 5f
r Periodic Trends • Atomic Radius • One half the distance between the nuclei of identical atoms that are bonded together
Na Mg Al Si P Atomic Radius Trend • Atomic Radius • Decrease across a period from left to right Going across the period the number of protons and the number of outer electrons increase Inner shell electrons remain the same Therefore the positive charge of the nucleus across the period has a greater pull on the outer electrons
Atomic Radius Trend • Atomic Radius • Increases down a group • Going down a group a new electron “shell” or level is added. As a level is added, the atom gets larger. ** Francium has the largest atomic radius
Ions • Ion: atom with an unbalanced charge (#p ≠ #e) • Cation: • positively charged atom • Lose electrons • Metals • Ex: Na1+, Mg 2+ • Anion: • negatively charged atom • Gain electrons • Nonmetals • Ex: Cl1-, O2-
Ionic Radii • same as atomic radii only with ions Period: • cation (metals) decrease left to right • anion (nonmetals) decrease left to right Group: • Increase down a group
Ionization Energy • The energy required to remove an electron from an atom • Forms cations • The higher the ionization energy the harder it is to remove an electron Element + energy A+ + e- Ex: Na + energy Na+ + e- Hint: Test Question
Ionization Energy Ionization Energy: Increases from left to right Increases from bottom to top ** Francium has the lowest ionization energy
Electron Affinity • The energy released when adding an electron to an atom • Creates anions Element + e- A- + energy Example: Cl + e- Cl1- + energy
Electron Affinity Electron Affinity: Increases from left to right Increases from bottom to top ** Fluorine has the highest electron affinity
Electronegativity • measure of the ability of an atom in a chemical compound to attract electrons • N, O, and the halogens are the most electronegative
Electronegativity Electronegativity: Increases from left to right Increases from bottom to top ** Fluorine has the highest electronegativity
Periodic Trend Summary Electronegativity Increases Electron Affinity Increases Ionization Energy Increases Atomic and Ionic Radius Increases Atomic and Ionic Radius Increases Electronegativity Increases Electron Affinity Increases Ionization Energy Increases