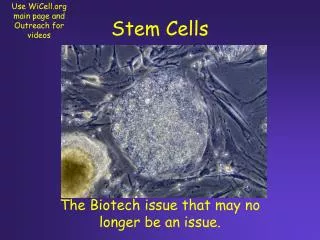
Stem Cells
Stem Cells. Keith Channon. Department of Cardiovascular Medicine University of Oxford John Radcliffe Hospital, Oxford. Adult Stem Cells. Unique cells that are capable of self-renewal Have the ability to differentiate through a committed lineage

Stem Cells
E N D
Presentation Transcript
Stem Cells Keith Channon Department of Cardiovascular Medicine University of Oxford John Radcliffe Hospital, Oxford
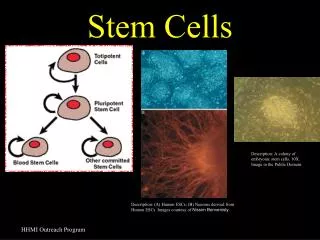
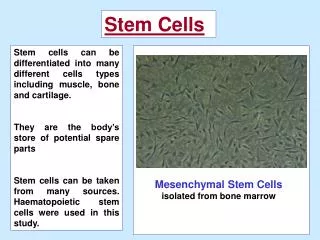
Adult Stem Cells Unique cells that are capable of self-renewal Have the ability to differentiate through a committed lineage Undergo further development within an adult organism v embryo They are multi(pluri)potent v totipotent
Phenotypically Characterised Adult Stem Cells N Engl J Med 2003;349:570-82.
Why are Stem Cells Relevant to Interventional Cardiology? Stem cells may offer new therapeutic approaches in cardiovascular disease Understanding stem cell biology challenges and informs our understanding of cardiovascular disease
Skeletal Myoblast Cell Transplant in Ischaemic Cardiomyopathy • Multinucleate • Negative for Connexin 43 Desmosomes Cadherin • No integration Menasché Pet al. Myoblast transplantationfor heart failure. Lancet 2001
Lin- c-kitPOS bone marrow cells from EGFP male mice to myocardium of female C57B6mouse • Injected in peri-infarct tissue 3-5hrs after LAD ligation Nature 2001;410:701-705
a c-kit neg c-kit pos Green=Cell Nuclei Red=Cardiac Myosin
Nature Medicine 2003;9:1195-1201 • CD117+ CD90+ CD34– MSCs from BM male rats isolated by adhesion to polystyrene, purified by immunoselection, • Transfected with murine Akt by VSV retrovirus • Permanent CAL of LAD in female rats • 60 mins post CAL MSCs injected 5 peri-infarct sites
Conclusions • Bone marrow-derived lineage negative progenitors regenerate infarcted murine myocardium • Autologous “skeletal lineage” progenitors improve cardiac function and survive in infarction scar
Stem Cells in Human Restenosis ? c-kit+ smc-actin Hibbert et al. Am J Physiol 2004
Stem Cells in Human Restenosis ? c-kit+ smc-actin Hibbert et al. Am J Physiol 2004
Nude mice • 1 day post femoral artery excision • Intracardiac medium, human µvascular ECs or EPCs
Endothelial Progenitor Cells- Role in Endothelial Maintenance Cytokines e.g. G-CSF RISK FACTORS high blood pressure high cholesterol / triglycerides smoking diabetes infections other STATINS EXERCISE Jonathan Hill, 2004
The STIMULATE Trial • TitleMulticenter, randomized controlled study of transplantation of bone marrow-derived progenitor cells into infarct vessels of patients following an acute myocardial infarction, acutely re-vascularised by percutaneous intervention. • Principal Investigators Prof. Dr. A. M. Zeiher & J.W. Goethe, University of Frankfurt • Sponsor Cardio-Cell, Zutphen, the Netherlands (parent Cryo Cell using subsid MainGen in Frankfurt ) • Monitoring CorTrial, Berlin • ObjectiveTo assess the safety and efficacy of intracoronary therapy with bone marrow-derived autologous progenitor cells with respect to improvement of myocardial function after an acute myocardial infarction treated by PTCA. • Design Multi-center,Randomized 1:1 • Primary endpointimprovement of left ventricular dysfunction at rest and during Dobutamine stress, assessed by echocardiography at 4 months.
THE PRIMATIVE Trial Leicester PercutaneousRandomisedInfusionof MarrowAspirateToImproveVentricularEfficiency • Principal Investigators Tony Gershlick, Nilesh Samani et al. • ObjectiveAssess the safety and efficacy of intracoronary therapy with bone marrow-derived autologous progenitor cells delivered at salvage PCI (DES) after acute MI, either early or late. • Design Single-center, Randomized, Placebo-Controlled n=150 • Primary endpointimprovement of left ventricular function, assessed by echocardiography and cardiac MRI at 4 months, and clinical events, up to 5 years.
Saline GM-CSF Saline GM-CSF
EPC Colony-Forming Capacity Following G-CSF J. Hill et al. JACC 2005; in press
G-CSF and In-stent Restenosis after MIKang et al. Lancet 2004 • Patients undergoing PCI with stenting of culprit artery following MI (3 days to 9 months) randomized to G-CSF ± apheresis / IC infusion, control • No AE’s associated with G-CSF treatment • At 6 month F/U, improved treadmill time, LVEF, SPECT perfusion in cell infusion group • In-stent restenosis determined in 7/10 G-CSF treated patients, 0/1 control • Study terminated
Isolation of EPCs for Stent Delivery vWF Flk-1 LDL-Uptake Shirota et al. Biomaterials 2003
Stem Cells in Interventional Cardiology cell therapy for post-MI repair vascular diseaserisk, biology,drug therapy cell therapy in PCI
Future Directions • Circulating or Bone Marrow Progenitor Cells? • Harvest or not, which subset? • Statins • Cytokine stimulation to release- e.g. CXCR4, new drugs • Circulating / Bone Marrow progenitor cells clinical trials- need to be blinded, placebo controlled, with hard end points • Understanding mechanisms remains critical to evaluating and targeting real benefits