Thermal conductivity
Thermal conductivity. Aim To be able to compare the thermal conductivities of a range of materials. Conduction. Hot. Cold. Direction of heat transfer.

Thermal conductivity
E N D
Presentation Transcript
Thermal conductivity Aim To be able to compare the thermal conductivities of a range of materials.
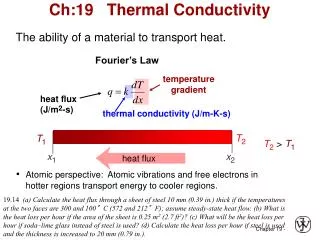
Conduction Hot Cold Direction of heat transfer Heat energy can be transferred by conduction. When materials are heated, the atoms vibrate more. This kinetic energy can be passed from atom to atom in conducting materials. Conduction happens in all materials, but in good conductors like metals, free electrons can transfer the heat energy more quickly. All metals are good conductors of heat. Insulators do not have free electrons and conduct heat poorly. Explain why metals always feel cold to the touch, whereas wood for example feels warm ? Conduction:
Equipment Materials, petroleum jelly, drawing pins, stop clock, safety glasses, tripod, heat mat, Bunsen burner Diagram
Method • Attach drawing pin, a measured distance from end of first material using petroleum jelly. • Place the material on a tripod • Heat the other end using a bunsen. • Time until petroleum jelly melts, and pin drops off. • Repeat twice with same material (cool it first) • Repeat with other materials
Risk Assessment Risk: Hazard: Control Measure:
Analysis Conductivity is…
Evaluation My experiment went… I did not have problems with… because… I had problems with … because… To improve my experiment I could… My results would be more reliable if I… I liked this experiment because… I disliked this experiment because…