Tissue Engineering
Tissue Engineering. James Zinckgraf. What is Tissue Engineering?. “Tissue Engineering is an interdisciplinary field that applies principles of engineering and life sciences towards the development of biological substitutes that aim to maintain, restore, or improve tissue function.” .

Tissue Engineering
E N D
Presentation Transcript
Tissue Engineering James Zinckgraf
What is Tissue Engineering? “Tissue Engineering is an interdisciplinary field that applies principles of engineering and life sciences towards the development of biological substitutes that aim to maintain, restore, or improve tissue function.”
Types of Tissue engineering • In Vivo • Manipulating cells while inside the body • In Vitro • Manipulating cells prior to implantation
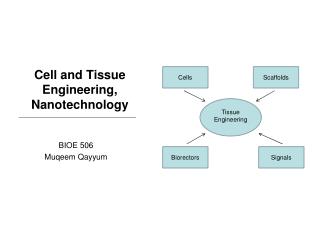
Components of Tissue Engineering • Cells • Matrix or Scaffold • Bioreactor • Cytokines and Growth Factors
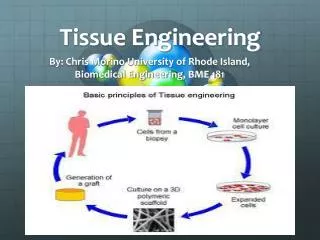
Cells • Cells are taken from the healthy tissue of what tissue is being created. • Skin cells for skin, bladder cells for bladder, etc. • Cells taken from donor or patient themselves • Autologous-donor and patient are the same • Allogenic-cells from another person • Xenogenic-cells from another species • Separated and screened for pathogens and disease • Placed onto scaffolds
Scaffolding • Act as Extra Cellular Matrix for cells • Function: Delivery of cells to desired sites, define space for formation, guide development with appropriate function • Needs to be able to control the structure and function of tissue in a predesigned manner. • Ideally they are biocompatible and biodegradable, without provoking inflammation or toxicity in the body.
Types of Scaffolding • Naturally derived • Collagen and alginate • Collagen is the most abundant and ubiquitous structural protein in the body. • Biologically recognized • Minimal inflammatory and antigenic responses • Alginate is from sea weed • Fibronectin • Encourages cell adhesion and growth
Types of Scaffolding • Accelular Tissue Matrices • Collagen rich • Formed from a segment of bladder or small intestine • Proven to support cell growth and regeneration for several tissues. • Polyesters • Naturally eliminated from body in form of C02 and H20 • Degradation rates in body can be manipulated • Lack biological recognition
Bioreactors • System where conditions are closely controlled to permit and induce a certain behavior in living cells or tissues • Provide controlled and steady flow of cell media • Factors necessary for cell growth: • pH, temp. pressure, nutrient supply, waste removal • Types of Bioreactors • Spinner Flasks, Rotating Vessels, Hollow Fiber, Perfusion reactors
Growth Factors • There are numerous growth factors for each tissue that can be engineered • Found naturally in body and help facilitate wound healing and cellular growth • Cytokines are widely used for multiple tissue types • Strings of amino acids that when attached to ECM initiate rapid multiplication of cells • Others include Epidermal Growth Factor (EGF), Fibroblast Growth Factor (FGF) and Platelet-Derived Growth Factor (PDGF) • All have many forms
Components of Human Skin • Dermis • Made of connective tissue, nourishes epidermis • Epidermis • Kertinocyte, melanocyte, a langerhans cells • Hypodermis • Barrier against physical harm, infection, dehydration.
Benefactors of Engineered skin • After injury or burns, body cannot act fast enough to manufacture replacement cells. • Before artificial skin burns covering 50% or more of body were considered fatal. • 2000, artificial skin cut death rates for people with 70%+ burns on their body from 100% to 40%. • Patients suffering from skin ulcers or recovering from surgery.
Process • Keratinocyte and Fibroblast cells collected from patient or donor skin. • Fibroblasts are added to a mix of collagen and sugar molecules that act as the scaffold. • Replicate dermal layer • Compound is brought to room temperature; collagen gels and traps fibroblasts. • Keratinocytes seeded onto new dermal tissue creating epidermal later. • After going through the bioreactor the keratinocytes are exposed to air and solidify.
History • Early as 6th century B.C. Hindu surgeons grafted skin from the body for nose reconstruction. • In 1907 Robert Hooke successfully grew ectodermal cells from a frog. • 1970, Dr. W.T. Hooke experimented growing and implanting cartilage into mice. • Tissue Engineering as a field was coined in 1987 by National Science Foundation
Future • Living tissues and organs can be easily and quickly assembled and integrated into the body to restore, replace, or enhance tissue and organ functions. • Organs such as the heart, lung, kidney and liver can be created in laboratories.
Sources Carlson, Bruce M. Principles of Regenerative Biology. Burlington: Elsevier, 2007. Chaudhuri, Julian, and Mohamed Al-Rubeai. Bioreactors for Tissue Engineering. Netherlands: Springer, 2005. Google Books. Web. Martin, Ivan, David Wendt, and Michael Heberer. "The role of bioreactors in tissue engineering." Trends in Biotechnology 22.2 (2004): 80-86. Web. McNicol, LoreAnne. "Reparative Medicine: Growing Tissues and Organs." Ed. Jean D. Sipe and Christine A. Kelley. The New York Academy of Sciences 961 (2002). Print Sachlos, E., and J.T. Czernuszka. "MAKING TISSUE ENGINEERING SCAFFOLDS WORK. REVIEW ON THE APPLICATION OF SOLID FREEFORM FABRICATION TECHNOLOGY TO THE PRODUCTION OF TISSUE ENGINEERING SCAFFOLDS." European Cells and Materials 5 (2003): 29-40. Web. 21 Feb. 2010. Metcalfe, Anthony D., and Mark W.J. Ferguson. "Tissue engineering of replacement skin: the crossroads of biomaterials, wound healing, embryonic development, stem cells and regeneration." UK Centre for Tissue Engineering (2006). Interface. Web. "Tissue Engineering." Wikipedia. Web. 15 Feb. 2010. <http://en.wikipedia.org/wiki/Tissue_engineering>.