1 / 27
Kinetic Theory of Gases: Basic Assumptions & Principles
270 likes | 304 Views
Explore the key assumptions of kinetic theory, such as large number of molecules and elastic collisions, to gain insight into gas behavior.
Download Presentation 

Kinetic Theory of Gases: Basic Assumptions & Principles
An Image/Link below is provided (as is) to download presentation
Download Policy: Content on the Website is provided to you AS IS for your information and personal use and may not be sold / licensed / shared on other websites without getting consent from its author.
Content is provided to you AS IS for your information and personal use only.
Download presentation by click this link.
While downloading, if for some reason you are not able to download a presentation, the publisher may have deleted the file from their server.
During download, if you can't get a presentation, the file might be deleted by the publisher.
E N D
Presentation Transcript
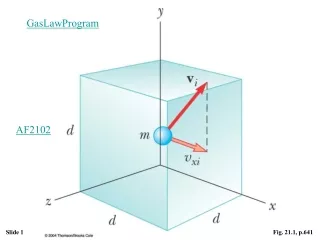
GasLawProgram AF2102 Fig. 21.1, p.641
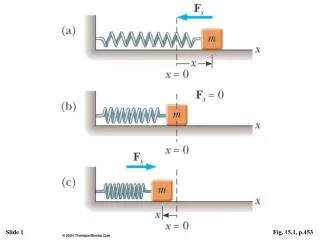
Basic Assumptions of the kinetic theory. • The number of molecules is Large. • The average separation is large compared to the size of each molecule • F=ma is valid • Molecules only interact via eleastic collisions at short range. • Molecules make elastic collisions with the walls • All molecules are identical.
AF2102 Fig. 21.2, p.642
AF2104 Fig. 21.4, p.647
http://hendrix.uoregon.edu/~dlivelyb/phys161/L12.html#adiabaticcomphttp://hendrix.uoregon.edu/~dlivelyb/phys161/L12.html#adiabaticcomp Fig. 21.5, p.650
AF21.12 AF21.11 Fig. 21.11, p.656
More Related