Molecular Orbitals
120 likes | 177 Views
Explore molecular orbitals, sigma and pi bonds, VSEPR theory, hybrid orbitals, and more to understand how atoms combine and form molecules. Learn about orbital hybridization, molecular shapes, and bond energies.

Molecular Orbitals
E N D
Presentation Transcript
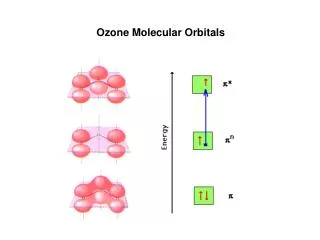
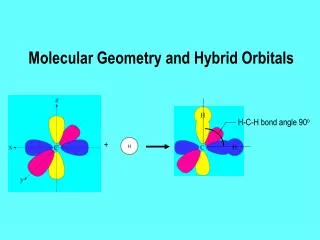
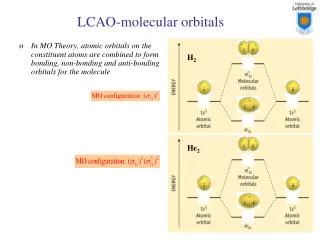
8.3 Molecular Orbitals • C. Bonding Theory: • 1. molecular orbitals: When two atoms combine, the molecular orbital model assumes that their atomic orbitals overlap to produce orbitals that apply to the entire molecule. • a. belongs to a molecule as a whole. • 2. bonding orbital: when a covalent bond is occupied by two electrons.
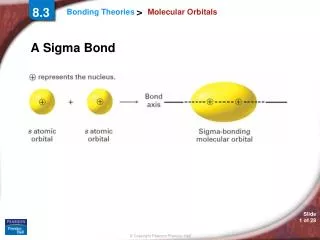
8.3 Molecular Orbitals • a. sigma bond: two atomic orbitals combine to form a molecular orbital that is symmetrical around the axis connecting two atomic nuclei. • b. pi bond ( bond): bonding electrons are found in sausage-shaped regions above and below the bond axis of the bonded atoms. • F F F--—-F
8.3 Molecular Orbitals • Pi-bonding Molecular Orbital
8.3 VSEPR Theory • 3. VSEPR (valence-shell electron-pair repulsion) theory: repulsion between electron pairs causes molecular shapes to adjust so that the valence-electron pairs stay as far apart as possible.
8.3 VSEPR Theory • a. Nine Possible Molecular Shapes
8.3 Hybrid Orbitals • Hybridization Involving Single Bonds
8.3 Hybrid Orbitals • Hybridization Involving Double Bonds
8.3 Hybrid Orbitals • Hybridization Involving Triple Bonds
Hybrid Orbitals • Simulation 7 Compare sp, sp2, and sp3 hybrid orbitals.
8.3 Section Quiz. • 1. A molecular orbital belongs to a • specific atom. • molecule as a whole. • specific pair of atoms. • central atom.
8.3 Section Quiz. • 2. VSEPR theory enables prediction of 3-dimensional molecular shape because the valence electron pairs • are attracted to each other. • form molecules with only four possible shapes. • stay as far apart as possible. • always form tetrahedral shapes.
8.3 Section Quiz. • 3. Orbital hybridization provides information about • both molecular bonding and molecular shape. • both molecular bonding and bond energy. • neither molecular bonding nor molecular shape. • neither molecular bonding nor bond energy.