CHAPTER 6 ELECTROLYSIS
330 likes | 539 Views
CHAPTER 6 ELECTROLYSIS. Electrolytes And Non-electrolyte Conductor A conductor is a substance which conducts electricity but is not chemically changed during the conduction . Insulators

CHAPTER 6 ELECTROLYSIS
E N D
Presentation Transcript
CHAPTER 6 ELECTROLYSIS
Electrolytes And Non-electrolyte Conductor A conductor is a substance which conducts electricity but is not chemically changed during the conduction . Insulators A non-conductor is a substance which does not allow the passage of electricity. Sometimes these non-conductors are used to protect something from electricity. They are then called insulators .
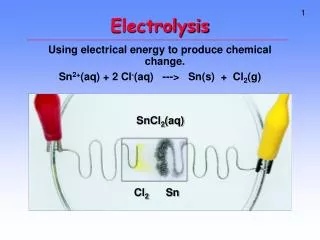
Electrolytes Electrolytes are compounds which when molten or dissolved in water conduct electric current and are decomposed in the process .
Non-electrolyte A non-electrolyte is a liquid which does not allow the passage of electricity.
Examples of non-electrolytes, weak electrolytes and strong electrolytes - Examples of electrolytes are acids,alkalis and salts dissolved in water or molten salts . - All these are ionic substances. - Solid ionic substances do not conduct electricity, as their ions are held together in fixed positions by strong forces . - In order for the ions to move, the solid must be dissolved in water or made molten, thereby destroying the ionic lattice .
There are 2 types of electrochemical cell, namely • 1.Electrolytic Cell • 2.Voltaic Cell (or sometimes is called Galvanic Cell) • - In electrolytic cell, electric current is flowed through an electrolyte to produce a chemical reaction. • In electrolytic cell, electrical energy is converted into chemical energy, and the process is called electrolysis. • In voltaic cell, chemical is used to produce electricity. • - In this cell, chemical energy is converted into electrical energy.
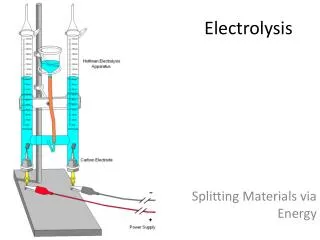
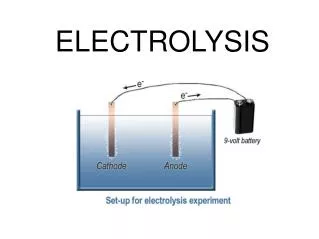
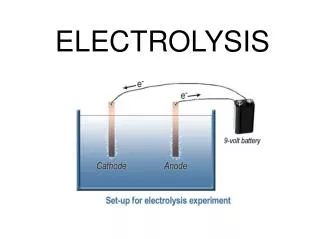
A suitable apparatus for electrolysis is shown in Figure above. - As we can see, the electrode connected to the positive terminal of the cell is positive electrode and is given a name, anode. - The electrode connected to the negative terminal of the cell is negative electrode and is called the cathode. - When electricity is passed through an electrolyte, chemical reaction happens. In this reaction, chemical is splitting up into 2 new substances. - All electrolytes are ionic, which means they are composed of positively and negatively charged ions. - On passing an electric current through the electrolyte, these ions move towards the oppositely charged electrode.
- Most negatively charged ions are non-metal ions, such as oxide (O2-, chloride (Cl-), Iodide (I-), etc. - During electrolysis, negatively charged ions move towards the positive electrode(anode). - The negative ions lose their electron(s) to the anode, which is positively charged. - The electron(s) is then move to the cathode through the external circuit (the wire). - The positively charged ions move towards the negative electrode(cathode'). - These positive ions are metal ions, such as copper (Cu2+), silver (Ag+), lead (Pb2+), etc, or hydrogen (H+). - At cathode, positive ions gain electron(s) from the cathode, which has an excess of electrons and therefore an overall negative charge.
This process results in the chemical decomposition of the electrolyte. It also allows electrons to travel from the cathode to the anode and hence allows conduction of electricity. During the electrolysis, electrical energy is supplied to the system to produce a chemical reaction. Therefore, during electrolysis, electrical energy convert into chemical energy.
This is composed of lead(II) ions, Pb2 + , and bromide ions, Br-. Its chemical formula is therefore PbBr2. A suitable apparatus which could be used to carry out this electrolysis is shown in Figure above. The bulb helps to show when electricity is flowing in the circuit, and until the lead(II) bromide is completely molten, the bulb does not light up . This confirms that electrolytes have to be molten for the ions to start to move to the electrodes and thereby conduct electricity.
In summary, the lead(II) bromide is split into its component elements : PbBr2 ---> Pb + Br2
Analysing the electrolysis of aqueous solutions Introduction - We have learnt that electrolyte can be molten ionic compound or aqueous solution of ionic compound, acid or alkali. - An aqueous solution is solution of water of a substance. For example, if you heat sodium chloride until it melts, it is called molten sodium chloride, but if you dissolve sodium chloride in water, it is called aqueous sodium chloride. - Electrolysis of aqueous solution is different from electrolysis of molten electrolyte. This is mainly because an aqueous solution contain more types of ions.
Let us take the example of molten sodium chloride and sodium chloride aqueous. • In molten sodium chloride, the ion present are sodium ion (Na+) and chloride ion (Cl-), due to the decomposition of the solid sodium chloride. NaCl ---> Na+ + Cl- • In sodium chloride aqueous, other than the decomposition of sodium chloride solid to form sodium and chloride ions, some of the water molecule will also disassociates to form hydrogen (H+) and hydroxide (OH-) ions. NaCl ---> Na+ + Cl- H2O ---> H+ + OH- • Which means in an aqueous solution, it can be more than 1 positive and negative ions. • When the ions move to the anode and cathode, only 1 negative ion and 1 positive ion will be selected to be discharged, and this is called selective discharge. • There are a few factors that determine which ion will be selected to be discharge, and this will be discussed in next section.
Factors Affecting Electrolysis There are three main factors that can affect the electrolysis products, there are: 1. position in the electrochemical series 2. the concentration and 3.the type of electrode
Electrochemical series - The chart above lists the ions in order of difficulty of discharge. -The ions at the top of the list is more difficult to be discharged, but as we go down the table, they become easier to be discharged. For example, Cu2+ easier to discharged compare with H+ and OH- is easier to be discharged compare with I-. - This series of ions is called the Electrochemical Series. -The lower the ion in the electrochemical series, the easier the ion to be discharged during electrolysis
As sulphuric acid is aqueous, it is composed not only of hydrogen ions (H+ ) and sulphate ions (SO42-), but also of hydroxide ions (OH-) from the water. H2SO4 + H2O --> 2H+ + SO42- + H+ + OH- • The apparatus used to carry out this electrolysis and collect the gases given off is shown in Figure 9 .8 . • When we have more than one type of ion moving to an electrode, selective discharge (or preferential discharge) takes place. • This means that the ion which can lose or gain electrons with the greatest ease is discharged, and the other ions, which are harder to discharge, remain in solution . • With the electrolyte aqueous sulphuric acid, migration of ions to the electrodes also occurs.
Concentration If the concentration of a particular ion is high, it will be selected to be discharged even though it is higher in the electrochemical series compares with another ion present in the solution. For example, if dilute hydrochloric acid is electrolysed, hydrogen gas is given off at the cathode and oxygen gas at the anode. However, when concentrated hydrochloric acid is electrolysed, hydrogen gas is still given off at the cathode, but chlorine rather than oxygen gas will be released at the anode, even though chloride is in a higher position in electrochemical series.
Electrolysis of Concentrated Sodium Chloride Solution (Brine)
The electrolytic cell used for electrolysis of concentrated sodium chloride solution is designed to collect gaseous products at both electrodes as shown in Figure above.
Type of Electrode This is best shown if we consider the electrolysis of aqueous copper(II) sulphate solution. Electrolysis of Copper(II) Sulphate by Using Carbon Electrode
Anode 1. If we use carbon electrodes, they are inert electrodes and do not affect the electrolysis. 2. Therefore, at the anode, we have a choice of sulphate or hydroxide ions . 3. The hydroxide ions are easier to discharge, so oxygen gas is given at the anode : Partial equation 40H- (aq) →O2(g) + 2H2O (l) + 4e- (oxygen gas given off) Cathode 1. At the cathode, we have a choice of copper or hydrogen ions . 2. The copper ions are easier to discharge, so we see a pink deposit of copper metal on the carbon electrode. Partial equation Cu2+ (aq) + 2e- → Cu (s) (copper metal deposited)
Electrolysis of Copper(II) Sulphate by Using Copper Electrode However, if we use copper electrodes, these are active electrodes and do affect the electrolysis.
Anode At the anode, the copper electrode dissolves into solution : Partial equation Cu(s) →Cu 2+ (aq) + 2e (copper electrode dissolves ) Cathode At the cathode, the copper ions are deposited as pink copper metal: Partial equation Cu2+ (aq) + 2e- →Cu (s) (copper metal deposited)