Trace elements analysis
Trace elements analysis. Elemental Analysis.

Trace elements analysis
E N D
Presentation Transcript
Elemental Analysis This chapter includes no detailed description of methods to determine individual mineral components. Such procedures are described in general textbooks of inorganic analysis, standard reference books on food analysis, and specialized textbooks on the determination of minerals in biological materials. The principles of instrumental methods used in the determination of mineral components and trace elements also were descried in previous chapters of this book. This chapter is primarily concerned with the applications of those principles to food analysis. Developments in the measurement of trace metal components in foods were described by LaFluer (1976), Winefordner (1976), Bratter and Schramel (1980), Das (1983), Schwedt (1984), and Benton-Jones (1984). Tshopel and Tolg (1982) reviewed the basic rules that have to be followed in trace analyses to obtain precise and accurate results at the nanogram and pictogram levels. These rules are as follows: 1. All materials used for apparatus and tools must be as pure and inert as possible. These requirements are only approximately met by quartz, platinum, glassy carbon, and, to a lesser degree,
polypropylene. 2. Cleaning of the apparatus and vessels by steaming is very important to lower blanks as well as element losses by adsorption. 3. To minimize systematic errors, microchemical techniques with small apparatus and vessels with an optimal ratio of surface to volume are recommended. All steps of the analytical procedure, such as composition, separation, preconcentration, and determination, are best done in one vessel (single-vessel principle). If volatile elements or compounds have to be determined, the system should be closed off and the temperature should be as low as possible. 4. Reagents, carrier gases, and auxiliary materials should be as pure as possible. Reagents that can be purified by subboiling point distillation are preferred. 5. Contamination from laboratory air should be avoided by using clean benched and clean rooms. By this, the blanks caused by dust can be decreased by at least two or three orders of magnitude.
6. Low and constant reaction temperature should be used. 7. Manipulations and different working steps should be restricted to a minimum in order to reduce unavoidable contamination. 8. All steps of the combined procedure should be monitored; this can best be done with radiotracers. 9. All procedures have to be verified by a second independent one or, even much better, by an interlaboratory comparative analysis. Element Enrichment The determination of trace element often requires enrichment of the elements, and/or the separation of many elements at the trace level from large amounts of major elements. Ion exchange has proved to be a valuable tool in the concentration, isolation, and recovery of ionic materials present in a solution in trace amounts. Ion-exchange chromatography on an ion-exchange resin can be also used for fractionation, separation, and the elimination of interfering ions.
Emission Spectroscopy Emission spectroscopy is the oldest instrumental method for trace analysis. It depends on observing and measuring the radiation emitted by atoms of the various means fall back to the original (or a lower) level. For each element there is a pattern of wavelengths characteristic of the element when excited in a particular way. By identifying the wavelengths in the spectrum, the sample can be analyzed. Emission spectroscopy is sensitive but the precision is rather low. Flame Photometry Early studies during the nineteenth century by J. F. Herschel, D. Alter, and G. kirchhoff and R. Bunsen laid the foundations for the qualitative differentiation of salts depending on their emission in a flame. Later researchers developed suitable techniques and instruments for quantitative analyses based on flame photometry. A modern flame photometer consists essentially of an atomizer, a burner, some means of isolating the desired part of the spectrum, a photosensitive detector, sometimes an amplifier, and finally a
method of measuring the desired emission by a galvanometer, null meter, or chart recorder (see Chapter 10 for details). The instruments are used primarily to determine calcium, sodium, and potassium. Atomic Absorption Spectroscopy Within the last two decades atomic absorption spectroscopy has found enthusiastic acceptance by science and industry. Hundreds of papers are published annually on basic research, instrumentation, specific analytical methods, and practical applications of atomic absorption spectroscopy. Atomic absorption spectroscopy is not quite as free from inter-element effects as was originally expected, but it is far better in this respect than any from of emission spectrography. It is quite sensitive; the limit of detection ranges from 0.01 ppm for magnesium to 5.00 ppm for barium; and the method is rapid (about 1000 determinations can be made per week). The equipment is relatively inexpensive (about $20000), only one-tenth the coast of X-ray fluorescence equipment. The limiting factor is the need for cathode lamps for each element or several combinations.
In atomic fluorescence spectroscopy, atoms are generated in the same way as in atomic absorption spectroscopy, expect that a cylindrical flame is used. The flame is irradiated by resonance radiation from a powerful spectral source, and the fluorescence that is generated in the flame is measured at right angles to the incident beam of radiation. This is done to minimize the contamination of the fluorescence signal by light from the source. Atomic absorption spectroscopy can be used in the ppm range; atomic fluorescence spectroscopy in the ppb range. Neutron Activation Analysis In neutron activation analysis, a weighed sample together with a standard that contains a known weight of the element sought is exposed to unclear bombardment. The radioactivity of the element in the sample is then compared with the radioactivity in the standard. Generally, a chemical separation is required to purify the radioisotopes of the element sought and to remove all other induced radioactivity. The quantity of the element in the sample is then calculated from the ratio of the separated activities. In some
instance, the final measurement of activity can be made on the intact sample. If the background remains inactive during nuclear bombardment or if the energies of the emitted radiations differ widely, a direct measurement of trace elements is possible. Also, if the trace element has a substantially longer half-life than the other induce activities, the interfering materials may be allowed to decay and the radioassay completed when the interference is insignificant. Results obtained by neutron activation generally are within 5% of the true value, and replicate analyses under favorable conditions are within 2-3% of the mean. The attractive features of neutron activation analyses are its wide applicability, high sensitivity, and satisfactory accuracy and precision. There have been numerous applications of activation analysis in botany and agriculture. X-Ray Spectroscopy There are three uses of X-rays in chemical analysis. Absorption methods are of limited practical because the adjustment of wavelength is most critical. X-ray diffraction is useful in crystallography and in establishing the complicated structure
of biological molecules. The use of X-ray for the identification of chemical components is based on emission methods, involving secondary or fluorescent emission. Measurement of the intensity and wavelength of fluorescence radiation is a well-established method of analysis. Coefficients of variation of about 1% in the concentration range 5-100% and of 5% in the range 0.1-1.0% can be obtained. In some instances determinations in the chemical combination of the element, and nondestructive in the sense that specimen examined is not destroyed, though some specimen preparation may be required. Instrumentation for X-ray spectroscopy is quite expensive. Glass Electrodes When a thin membrane of glass is interposed between two solutions, an electrical potential difference is observed across the glass. The potential depends on the ions present in the solutions. Depending on the composition of the glass, the response may be primarily to the hydrogen ion, to other cations, or even to organic cations. The electrodes are unaffected by oxidants and reducing agents, and only slightly affected by anions (except fluoride) or by
high concentrations of proteins and amino acids. Miscellaneous Methods Trace elements are determined in many laboratories by specific colorimetric and turbidimetric methods, by fluorescence analysis, and by polarography. The use of infrared spectroscopy in determining polyatomic ions was described by Miller and Wilkins. Relatively simple chromatographic methods for rapid routine evaluation of trace elements in crops and foods were described by Duffield and Coulson. Impressive advances have been made in developing instruments that permit an essentially complete elemental analysis to be performed in situ on the structures observed in the tissues of thin sections prepared by standard histological methods. The electron probe microanalyzer or electron probe X-ray scanning microscope can perform nondestructive elemental chemical analyses on localized regions with diameters as small as 1μm and volumes of a few cubic micrometers. The limit of delectability is about 0.1%, and many inorganic elements can be measured.
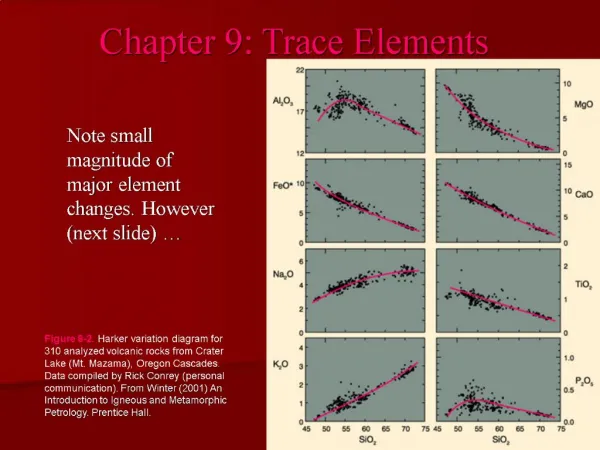
Another promising technique that has been adapted to microanalysis of inorganic elements is the laser microprobe. In this instrument, a laser beam is flashed through the optics of a regular microscope set to analyze a very small arc. The instrument is attached to a sensitive spectrograph. Finally, mention should be made of biological methods of trace analysis. Comparison of Methods Bowen described the results of elemental analyses of a standard plant material analyzed for 40 elements by 29 laboratories. The techniques used were neutron activation analysis, atomic absorption spectroscopy, a catalytic technique, colorimetry, flame photometry, turbidimetry, and titrimetric analysis. Consistent results were obtained by more than one laboratory for Au, B, Br, Ca, Cl, Co, Cr, Fe, Ga, I, Mn, Mo, N, P, Rb, S, Sc, and W. Small differences in results were obtained by different techniques were found for Cu, K, Mg, Na, P, Se, Sr, and Zn. For example, flame photometry gave high results for sodium, activation analysis without chemical separation was unreliable for determining potassium and magnesium, and atomic absorption spectrometry gave high
result for copper and strontium. Gross discrepancies were found in the result reported for aluminum, arsenic, mercury, nickel, and titanium. The significance of databases and food composition compilations in trace element analyses was stressed by Southgate. According to Wolf and Hamly, inorganic trace elements of interest in human health can be divided into those that are of nutritional and toxic interest, those that are primarily of nutritional interest, those that are primarily of toxic interest. The two techniques considered by the authors as having the required sensitivity and greatest potential for accurate trace element analysis are atomic spectroscopy and neutron activation analysis. Hocquellet determined cadmium, lead, arsenic, and tin in vegetable and fish oils by atomic absorption with electrothermal atomization by an oven equipped with a graphite tube. Addition of dithiocarbamate for cadmium or of dithiocarbamate for lead and arsenic decreased volatility of the elements. Detection limits of 0.5-3.0ng/g and satisfactory recoveries were obtained in the 20-ppb range when samples of oil diluted in chloroform or in methylisobutyl ketone were injected into the atomizer. This rapid (minutes
compared to hours for methods with nitrosulfuric digestion) direct determination method was recommended for rapid routine testing of large numbers of samples. Noller and Bloom described an integrated analytical scheme for the determination of major (sodium, potassium, calcium, and magnesium) and minor (zinc, copper, nickel, iron, chromium, cesium, lead, tin, and mercury) elements in foods. The methods involved flame atomic absorption and flame emission spectrometry for all elements expect mercury, for which flameless atomic absorption was recommended. In a collaborative study involving 13 Australian laboratories, cadmium, copper, iron, lead, tin, and zinc were determined in spiked and unspiked samples of apple puree. Atomic absorption was used in the flame mode to determine copper, iron, and zinc; it was efficient and accurate and yielded low interlaboratory coefficients of variation and good recoveries. However, tin many be lost in ashing as volatile stannic chloride or as insoluble metastannic acid. Lead was determined by direct flame atomic absorption, by solvent extraction followed by flame atomic absorption, and by electrothermal atomization. The methods yielded comparable results.
The graphite furnace as an alternative to the combustion flame in atomic absorption spectrometry (AAS) because available commercially about 1970. Electrothermal atomization offers many advantages in terms of sensitivity and smaller sample requirements. At the same time, the comparatively long analysis times, the need to optimize conditions for each element, and the occurrence of matrix interferences impaired the application of graphite furnace AAS in routine analysis. Recent developments in instrument design and methodology have reduced the severity of those problems, and graphite furnace-AAS is the most widely used method for determination of trace elements. A combination of wet charring and dry ashing suitable for the determination of trace metals in oily foods by graphite furnace-AAS was described by Seong Lee et al. BACK