Diffraction and Crystal Structure
Diffraction and Crystal Structure. NANO Workshop 2006. Outline. Light and X-rays Diffraction basics Determining crystal structures Activities!. What is Light?. Particle or wave? Both! Photons: particles or “quanta” of light E = hf Many photons → wavelike

Diffraction and Crystal Structure
E N D
Presentation Transcript
Diffraction and Crystal Structure NANO Workshop 2006
Outline • Light and X-rays • Diffraction basics • Determining crystal structures • Activities!
What is Light? • Particle or wave? Both! • Photons: particles or “quanta” of light E = hf • Many photons → wavelike • A disturbance that propagates • Usually requires a medium
Anatomy of a Wave • Wavelength • Frequency f • # of cycles per unit time • Speed v = f • Amplitude A
Electromagnetic Waves • Speed = 3 105 km/sec (about 186,000 mi/sec)
Visible Light • The color of visible light is determined by its wavelength • White light is a mixture of all colors • We can separate out individual colors with a prism
Visible Light 400–440 nm Violet 440–480 nm Blue 480–530 nm Green 530–590 nm Yellow 590–630 nm Orange 630–700 nm Red
Superposition and Interference • When multiple waves pass through the same place, the total wave is obtained by adding together the individual wave displacements (Principle of Superposition)
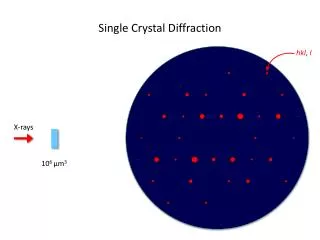
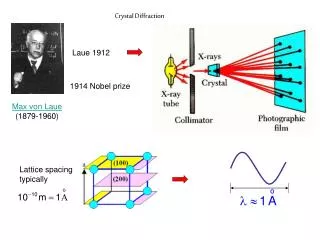
X-Ray Diffraction • X-rays have wavelengths comparable to atomic sizes and spacings, about 10–10 m • Crystals and molecules reflect X-rays in specific patterns depending on their structures X-ray diffraction pattern of myoglobin
Interaction of X-Rays with Atoms • Involves the electrons, primarily
1 2 d Fraunhofer Diffraction • “Fraunhofer” = incident, outgoing rays parallel • Start with single plane of atoms (a grating) • Wave “in phase” – incident crests and troughs aligned
Fraunhofer Diffraction • Extra distance traveled by 2 is d sin • If this is an integer number of wavelengths, we get constructive interference 1 d 2
Condition for Maxima where n = 0, 1, 2, … • If is known, measuring tells us d • Or if d is known we can get , etc.
Many Diffraction Centers • All combine constructively at the specified angles • Nice sharp maxima!
Bragg’s Law • W. H. Bragg and W. L. Bragg, 1913 (Nobel 1915) • Condition for constructive interference: • Diffraction from different sets of planes in the crystal gives a picture of the overall structure
More Information • Intensities of diffraction maxima can vary – more information about detailed structure • Symmetry of the crystal structure is reflected in the diffraction pattern
Electron Diffraction • Can be done with particles too, due to their wave nature! • Direct test of the De Broglie relation = h/p (Davisson and Germer, Thomson) Electron diffraction image
Optical Transform Exercises • Uses visible light, ~ 400-700 nm • Diffracting patterns created with features on this scale • Illuminate with laser pointer – coherent light source
Discovery Slide • Horizontal line patterns (a and c) • Diffraction pattern is a set of vertical dots – why? • Check spacing of dots for different line spacings • “Reciprocal lattice effect” • Check spacing of dots for different wavelengths
Group Exercise • Given a known wavelength of light (it’s shown on the laser pointer), determine the line spacing of the Discovery Slide pattern a.
Other Exercises • Explore the symmetry relations between diffraction patterns and the “atomic” arrangements • Look for variations in intensity for diffraction maxima – when does this occur?