BACKGROUND
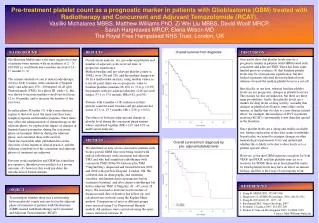
RESULTS. BACKGROUND. DISCUSSION. Overall survival from diagnosis. Glioblastoma Multiforme is the most aggressive form of primary brain tumour, with an incidence of 2 – 5/ 100 000/ yr worldwide and a median survival of 14 – 17 months (1, 2).

BACKGROUND
E N D
Presentation Transcript
RESULTS BACKGROUND DISCUSSION Overall survival from diagnosis Glioblastoma Multiforme is the most aggressive form of primary brain tumour, with an incidence of 2 – 5/ 100 000/ yr worldwide and a median survival of 14 – 17 months (1, 2). The current standard of care is radical radiotherapy (60 Gy/ 30 #/ 6 weeks) with concurrent (75mg/m2 daily) and adjuvant (150 – 200 mg/m2 d1-d5 q28) Temozolamide (TMZ). In a phase III study (1), this was shown to increase median overall survival from 12 to 14 months, and to increase the number of late survivors. In earlier phase II studies (3) with a near-identical regime to that now used, the main toxicities were lymphocytopenia and thrombocytopenia. Since these may affect the administration of chemotherapy in the adjuvant phase, we explored the impact of changes in haematological parameters during the concurrent phase on treatment delivery during the adjuvant phase, and correlated these with survival.There has been little other published data on the outcomes of this regime in clinical practice, and the differing contribution of the concurrent and adjuvant phases of treatment are unknown. Previous work on platelets and GBM has found that pre-operative thrombocytosis predicts for a poorer outcome (4); however, this work pre-dates the introduction of Temozolamide. On univariate analysis, sex, pre-adjuvant platelets and number of adjuvant cycles received were of prognostic significance. Median baseline and pre-adjuvant platelet counts (x 109/L) were 256 and 216, and the median change was 38. In a multivariate analysis, using median values as a cut-off point, there was no prognostic value to baseline platelets (median OS 19.6 vs. 17.6, p = 0.95) but patients with pre-adjuvant platelets <216 survived longer (33.1 vs 17.6 months; HR=0.42; p=0.03). Patients with a smaller (<38) reduction in their platelet count between baseline and pre-adjuvant did worse (17.6 vs. 25.5 months; HR = 2.58, p = 0.02). The effects of both pre-adjuvant and change in platelet level during the concurrent phase remain when considered together (HR = 2.07 and 0.53 on multivariate analysis). Our results show that platelet levels may be a prognostic marker in patients with GBM treated with concurrent and adjuvant TMZ. There has been some limited previous evidence (4) that baseline platelet levels may be of prognostic significance, but this looked at patients who had elevated platelet levels, whereas we used the median platelet level as a cut-off. Specifically, in our data, whereas baseline platelet levels are not prognostic, changes in platelet level are. The reasons for this are unknown, but there are three main possibilities: firstly, that platelet levels are a marker for drug levels or drug activity, secondly that changes in platelet level lead to some effect on the tumour, or thirdly that it is due to a non-disease related effect: for example, the incidence of DVT in patients receiving RCAT is substantially lower than that quoted in the literature. Since platelet levels are a cheap and readily available test, further exploration of this data seems worthwhile. In particular, we intend to examine changes in other haematological parameters to try and understand whether this is likely to be due to dose levels or is a platelet specific effect. However, given that GBM expresses receptors for both VEGF and PDGF, and that platelets may act as a reservoir for PDGF, there are at least plausible routes by which platelet levels may have an effect on tumour biology, and this is the focus of our ongoing work. METHOD Overall survival from diagnosis by pre- adjuvant platelet level We identified seventy-seven consecutive patients with biopsy-proven GBM who were being treated with radical radiotherapy with concurrent and adjuvant TMZ and who had completed radiotherapy with concurrent TMZ (60Gy/30 fractions plus TMZ 75mg/m2/day), diagnosed and treated between 2002 and 2009 at Royal Free Hospital , London, UK. We collected data on demographic and treatment variables, and haematological parameters before treatment (baseline) and after chemo-radiotherapy but before adjuvant TMZ (150mg/m2, d1 – d5, every 28 days). We assessed overall survival from date of diagnosis until date of death or last follow up, and calculated survival rates using the Kaplan-Meier method. Comparisons of rates in different groups were assessed using Cox Proportional Hazards models. All analyses were carried out using the open-source statistical software, R. REFERENCES OBJECT 1. Stupp R, NEJM 2005, 352:987-996 2. Hargreaves S, ASTRO Proceedings, 2010, Abs.No.912 3. Stupp R, JCO 2002, 20: 1375 - 82 4. Brockmann MA, Neuro-Oncology, 2007 4. Stommel J, Science 2007, 318:287-290 4. Furnari F, Genes & Development 2007, 21:2683-2710 Contact: Matt.Williams@nhs.net To investigate whether there was a relationship between platelet counts and survival in the adjuvant phase of treatment of patients with Gliobastoma (GBM) treated with Radiotherapy and Concurrent and Adjuvant Temozolamide (RCAT).